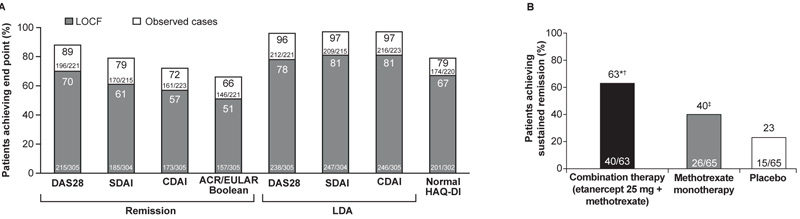
Fig. (1)
(A) The proportion of patients receiving etanercept 50 mg plus methotrexate in the open-label mITT population who achieved clinical and functional end points at the final visit of the open-label phase (LOCF, observed cases). All comparisons, p<0.0001 versus baseline. (B) The proportion of patients randomized to reduced-dose (25 mg) etanercept plus methotrexate combination therapy, methotrexate monotherapy, or placebo in the double-blind mITT population who achieved sustained remission in the double-blind period by randomized treatment group (NRI). *p<0.01 combination versus methotrexate monotherapy; †p<0.0001 combination versus placebo; ‡p<0.02 methotrexate monotherapy versus placebo (mITT). Observed cases = completers. ACR/EULAR: American College of Rheumatology/European League Against Rheumatism; CDAI: Clinical Disease Activity Index; DAS28: Disease Activity Score for 28-joint counts; LDA: Low Disease Activity; LOCF: Last Observation Carried Forward; mITT: modified Intent-To-Treat; NRI: Nonresponder imputation; SDAI: Simplified Disease Activity Index.