- Home
- About Journals
-
Information for Authors/ReviewersEditorial Policies
Publication Fee
Publication Cycle - Process Flowchart
Online Manuscript Submission and Tracking System
Publishing Ethics and Rectitude
Authorship
Author Benefits
Reviewer Guidelines
Guest Editor Guidelines
Peer Review Workflow
Quick Track Option
Copyediting Services
Bentham Open Membership
Bentham Open Advisory Board
Archiving Policies
Fabricating and Stating False Information
Post Publication Discussions and Corrections
Editorial Management
Advertise With Us
Funding Agencies
Rate List
Kudos
General FAQs
Special Fee Waivers and Discounts
- Contact
- Help
- About Us
- Search

The Open Cell Signaling Journal
(Discontinued)
ISSN: 1876-3901 ― Volume 4, 2012
Dual Philosophy in Death Receptor Signalling
Chahrazade Kantari*, Henning Walczak
Abstract
Tumour necrosis factor (TNF) is the founding member of a cytokine family with important roles in both, physiology and pathological conditions. The two seemingly opposing cellular responses to stimulation by TNF itself are death and induction of pro-inflammatory signalling. TNF and other TNF superfamily (SF) members signal by crosslinking their cognate receptors. These form part of the TNF receptor SF (TNFRSF). Members of this family have between two and six characteristic cysteine-rich repeats in their extracellular domain. These repeats are crucial for receptor-ligand interaction. Members of the TNFRSF come in three flavours: as type I transmembrane proteins, attached to the plasma membrane by a glycosyl¬phosphatidyl¬inositol (GPI) anchor, or as secreted soluble proteins. The latter receptors act as decoys for their respective ligands. To date 30 members of the TNFRSF are known. Six of them form part of the subfamily of the death receptors. Death receptors are characterised by the presence of an intracellular death domain (DD). Amongst the death receptors there are again at least two subclasses, the ones which recruit the Fas-Associated Death Domain (FADD) and the ones that recruit the TNFR-Associated Death Domain (TRADD) protein. The primary function of FADD-recruiting receptors is to induce apoptosis whilst the primary function of the TRADD recruiters is to activate pro-inflammatory signalling (Fig. 1). However, from a second platform both systems are also capable of triggering the respective other signalling outcome
The six human DD-containing receptors,
TNF-R1 (p55/p60 TNF-R), CD95 (Fas, APO-1), death receptor 3 (DR3, TRAMP), TRAIL-R1 (DR4), TRAIL-R2 (DR5)
and DR6 (TNFRSF21) are activated by their respective ligands: TNF, CD95L (FasL/APO-1L), TL1A, TRAIL (Apo2L),
and a specific amino-terminal cleavage fragment of the β-amyloid precursor protein (APP), N-APP. They are
transmembrane proteins which contain repeats of 2-4 cysteine-rich domains (CRDs) in the extracellular portion required
for ligand binding and an intracellular death domain (ICD) capable of recruiting specific adaptors proteins. Whilst the
primary signal output of the TRADD-recruiting ICDs of TNF-R1 and DR3 (shown in dark grey) is the activation of
inflammatory signalling, the FADD-recruiting ICDs of CD95 and the TRAIL death receptors (shown in light grey)
induce apoptosis as their primary signalling output.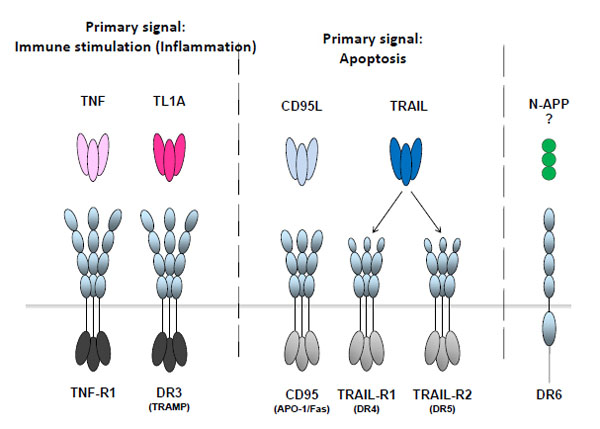
The 6 human DD-containing receptors and their known ligands.
Article Information
Identifiers and Pagination:
Year: 2011Volume: 3
First Page: 27
Last Page: 34
Publisher Id: TOCELLSJ-3-27
DOI: 10.2174/1876390101103010027
Article History:
Received Date: 2/5/2010Revision Received Date: 21/8/2010
Acceptance Date: 22/10/2010
Electronic publication date: 14/4/2011
Collection year: 2011
open-access license: This is an open access article licensed under the terms of the Creative Commons Attribution Non-Commercial License (http: //creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted, non-commercial use, distribution and reproduction in any medium, provided the work is properly cited.
* Address correspondence to this author at the Tumour Immunology Unit, Department of Medicine, Imperial College London, Hammersmith Hospital Campus, 10th floor, Commonwealth Building, London W12 0NN, UK; Tel: +44-20-8383 2094; E-mail: c.kantari@imperial.ac.uk
| Open Peer Review Details | |||
|---|---|---|---|
| Manuscript submitted on 2-5-2010 |
Original Manuscript | Dual Philosophy in Death Receptor Signalling | |
