- Home
- About Journals
-
Information for Authors/ReviewersEditorial Policies
Publication Fee
Publication Cycle - Process Flowchart
Online Manuscript Submission and Tracking System
Publishing Ethics and Rectitude
Authorship
Author Benefits
Reviewer Guidelines
Guest Editor Guidelines
Peer Review Workflow
Quick Track Option
Copyediting Services
Bentham Open Membership
Bentham Open Advisory Board
Archiving Policies
Fabricating and Stating False Information
Post Publication Discussions and Corrections
Editorial Management
Advertise With Us
Funding Agencies
Rate List
Kudos
General FAQs
Special Fee Waivers and Discounts
- Contact
- Help
- About Us
- Search

Open Chemistry Journal
(Discontinued)
ISSN: 1874-8422 ― Volume 8, 2021
Synthesis and In-vitro Antifungal Evaluation of 5- Pyrazolones
Poonam Gupta*, Jitendra K. Gupta
Abstract
A series of 4-chloro-3-methyl-N-(substituted phenyl)-5-oxo-4,5-dihydro-1H-pyrazole-1- carbothioamide were synthesized using earmark reaction of thiosemicarbazides and ethyl-2-chloroacetoacetate in DMF. All structures of the synthesized compounds were distinguished on the basis of IR, 1H-NMR, 13C-NMR and also elemental analysis. Synthesized Compounds were examined their potent antifungal activity using disc diffusion technique against three fungal pathogens viz Aspergillus niger, Candida albicans and Curvularia. Fluconazole were used as standard drug respectively. All compounds exhibited good to moderate activity.
Article Information
Identifiers and Pagination:
Year: 2016Volume: 3
First Page: 17
Last Page: 24
Publisher Id: CHEM-3-17
DOI: 10.2174/1874842201603010017
Article History:
Received Date: 09/04/2015Revision Received Date: 27/7/2015
Acceptance Date: 9/10/2015
Electronic publication date: 09/05/2016
open-access license: This is an open access article licensed under the terms of the (https://creativecommons.org/licenses/by/4.0/legalcode), which permits unrestricted, noncommercial use, distribution and reproduction in any medium, provided the work is properly cited.
* Address correspondence to this author at the Research Scholar Jiwaji University, Gwalior, India; Tel: 9039926286; E-mail: poonamgupta_001@yahoo.com
| Open Peer Review Details | |||
|---|---|---|---|
| Manuscript submitted on 09-04-2015 |
Original Manuscript | Synthesis and In-vitro Antifungal Evaluation of 5- Pyrazolones | |
INTRODUCTION
In recent years, fungal infections spreaded worldwide infection to life threatening systemic ailments involving the internal organs ranging from minor superficial skin and mucous membrane. The search for new, effective and safe nuclei leads to important modifications in the existing drugs by increasing their efficacy as well as formulating new bioactive agents by molecular modifications. The role of medicinal chemistry is essential and sustainable for previous and current generation. In the practice of medicinal chemistry developed from an empirical organic synthesis of new compound based on the modification of structure and identifies their biological activity [1William, D.A.; Lemke, T.L. Principles of Medicinal Chemistry; Williams and Wilkins: Lippincott, 2002, vol. 5, p. 36., 2Pandeya, S.N. A Text Book of Medicinal Chemistry; SG Publisher: Singapore, 2004, vol. 1, pp. 2-3.]. Moreover, various heterocyclic and biologically active compounds have five-member nitrogen, sulphar, oxygen containing heterocyclic ring [3Lednicer, D.; Mitscher, L.A. In Organic Chemistry of Drug Synthesis; Wiley Interscienc: New York, 1997, vol. 1, p. 226.]. Pyrazolone is a five membered lactum ring, containing two nitrogen and one ketonic group in its structure.
Pyrazolones have acquired versatile importance as drug substances in pharmaceutical industry of their biological importance. For instance, various pyrazolones drugs, viz. phenazone, propyphenazone, ampyrone and metamizole are useful antipyretic and analgesic drugs [4Himly, M.; Jahn-Schmid, B.; Pittertschatscher, K.; Bohle, B.; Grubmayr, K.; Ferreira, F.; Ebner, H.; Ebner, C. Ig E-mediated immediate-type hypersensitivity to the pyrazolone drug propyhenazone. J. Allergy Clin. Immunol., 2003, 111, 882-888.
[http://dx.doi.org/10.1067/mai.2003.163] [PMID: 12704373] ]. Therefore, pyrazolones possess antimicrobial, antifungal [5Al-Haiza, M.A.; El-Assiery, S.A.; Sayed, G.H. Synthesis and potential antimicrobial activity of some new compounds containing the pyrazol-3-one moiety. Acta Pharm., 2001, 51, 251-261.], anti-mycobacterial [6Castagnolo, D.; Manetti, F.; Radi, M.; Bechi, B.; Pagano, M.; De Logu, A.; Meleddu, R.; Saddi, M.; Botta, M. Synthesis, biological evaluation, and SAR study of novel pyrazole analogues as inhibitors of Mycobacterium tuberculosis: part 2. Synthesis of rigid pyrazolones. Bioorg. Med. Chem., 2009, 17(15), 5716-5721.
[http://dx.doi.org/10.1016/j.bmc.2009.05.058] [PMID: 19581099] , 7Radi, M.; Bernardo, V.; Bechi, B.; Castagnolo, D. Microwave-assisted organocatalytic multicomponent Knoevenagel/hetero Diels-Alder reaction for the synthesis of 2,3-dihydropyran[2,3-c]pyrazoles. Tetrahedron Lett., 2009, 50, 6572-6575.
[http://dx.doi.org/10.1016/j.tetlet.2009.09.047] ], antibacterial [8Moreau, F.; Desroy, N.; Genevard, J.M.; Vongsouthi, V.; Gerusz, V.; Le Fralliec, G.; Oliveira, C.; Floquet, S.; Denis, A.; Escaich, S.; Wolf, K.; Busemann, M.; Aschenbrenner, A. Discovery of new Gram-negative antivirulence drugs: structure and properties of novel E. coli WaaC inhibitors. Bioorg. Med. Chem. Lett., 2008, 18(14), 4022-4026.
[http://dx.doi.org/10.1016/j.bmcl.2008.05.117] [PMID: 18571407] ], anti-inflammatory [9Sauzem, P.D.; Machado, P.; Rubin, M.A.; da S Sant’anna, G.; Faber, H.B.; de Souza, A.H.; Mello, C.F.; Beck, P.; Burrow, R.A.; Bonacorso, H.G.; Zanatta, N.; Martins, M.A. Design and microwave-assisted synthesis of 5-trifluoromethyl-4,5-dihydro-1H-pyrazoles: novel agents with analgesic and anti-inflammatory properties. Eur. J. Med. Chem., 2008, 43(6), 1237-1247.
[http://dx.doi.org/10.1016/j.ejmech.2007.07.018] [PMID: 17889969] ], antitumor [10Pasha, F.A.; Muddassar, M.; Neaz, M.M.; Cho, S.J. Pharmacophore and docking-based combined in-silico study of KDR inhibitors. J. Mol. Graph. Model., 2009, 28(1), 54-61.
[http://dx.doi.org/10.1016/j.jmgm.2009.04.006] [PMID: 19447057] ], gastric secretion stimulatory [11Rosiere, C.E.; Grossman, M.I. An analog of histamine that stimulates gastric acid secretion without other actions of histamine. Science, 1951, 113(2945), 651-653.
[http://dx.doi.org/10.1126/science.113.2945.651] [PMID: 14845699] ], anti-depressant [12Bailey, D.M.; Hansen, P.E.; Hlavac, A.G.; Baizman, E.R.; Pearl, J.; DeFelice, A.F.; Feigenson, M.E. 3,4-Diphenyl-1H-pyrazole-1-propanamine antidepressants. J. Med. Chem., 1985, 28(2), 256-260.
[http://dx.doi.org/10.1021/jm00380a020] [PMID: 3968690] ] and antifilarial activities [13Chauhan, P.M.; Singh, S.; Chatterjee, R.K. Antifilarial profile of substituted pyrazoles: a new class of antifilarial agents. Indian J. Chem. Sect. B, 1993, 32, 858-861.]. Many attempts have been made to synthesize, characterize and to study biological activity of pyrazolones [14Shestopalov, A.M.; Emelyanova, Y.M.; Shestopalov, A.A.; Rodinovskaya, L.A.; Niazimbetova, Z.I.; Evans, D.H. Cross of substituted 6-amino-spiro-4-(N-R-piperidine-4’)-2H,4H-pyrano[2,3-condensation of derivatives of cyanoacetic acid and carbonyl compounds. Part 1. One-stage synthesis c]pyrazole-5-carbonitriles. Tetrahedron, 2003, 59, 491-7496.
[http://dx.doi.org/10.1016/S0040-4020(03)01178-5] ]. Interest in the chemistry of a new organic photochromic compound containing pyrazolone-ring was synthesized and characterized by Liu et al. They evaluated their photocromic properties were related to the photoisomerization and time-dependent UV-vis spectra, and these compounds exhibited good antibacterial activities [15Guangfei, L.; Lang, Li.; Dianzeng, J.; Kaibei, Yu. Synthesis, structure and photochromic properties of 1-phenyl-3-methyl-4-(4-bromobenzal)-pyrazolone-5 thiosemicarbazone. Struct. Chem., 2005, 16, 135-140.
[http://dx.doi.org/10.1007/s11224-005-2837-6] ]. Atudosie et al. [16Baciu-Atudosie, L.; Ghinet, A.; Belei, D.; Gautret, P.; Rigo, B.; Bicu, E. An efficient one-pot reaction for the synthesis of pyrazolones bearing a phenothiazine unit. Tetrahedron Lett., 2012, 53, 6127-6131.
[http://dx.doi.org/10.1016/j.tetlet.2012.08.152] ] have been reported the synthesis of new 5- substituted-2-[2-(2-substituted-10H-phenothiazin-10-yl)-2-oxoethyl]-2,4-dihydro-3H-pyrazol-3 one containing phenothiazine unit by reaction of N-chloroacetyl compound, ethyl acetoacetate with hydrazine hydrate and their were evaluated antiproliferative activity. In the recent years, the chemistry and antibacterial activity of pyrazolone have been investigated and synthesized to be novel pyrazolones from easily available starting materials and their broad range of antimicrobial and anti-inflammatory activity were evaluated [17 Anonymous. British Pharmacopoeia; H. M. S. Publication Center: UK, 1988, vol. 2, p. A205.-23Vijesh, A.M.; Isloor, A.M.; Isloor, S.K.; Shivananda, K.N.; Shyma, P.C.; Arulmoli, T. Synthesis of some new pyrazolone derivatives as potent antimicrobial agents. Der. Pharm. Chem., 2011, 3, 454-463.]. The study was aimed at exploring our synthesis of some new biologically active pyrazolone derivatives by the reaction of thiosemicarbazide and ethyl-2-chloro acetoacetate.
MATERIALS AND METHODS
All materials were of commercial reagent grade and purchased from Sigma-Aldrich. All reaction were monitored by column chromatography with E-Merck silica precoated plates and visualization was executed by ultraviolet and iodine chamber. Melting points were measured by open glass capillary and are uncorrected. 1H and 13C NMR spectra in CDCl3 were taped on a BRUKER AVANCE II 129 400 MHz FT-NMR spectrometer (Bruker Bio Spin, Switzerland) at 400 and 100 MHz, respectively. IR spectra were taped using Perkin Elmer FTIR spectrophotometer. Elemental analysis was performed on Elementarvario MICRO cube CHN analyser. All yields refer to isolated products.
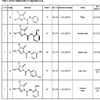
Literature revealed that more possibilities to finding a suitable derivative, which would express their activity more than already known drugs containing N-N and semicarbazone moiety. With the alarming trends in bacterial resistance to many pyrazolone derivatives it has become necessary to synthesize some novel pyrazolone for bioassay of antifungal activity and the need for drugs with more specific antifungal activity. Therefore it was considered of interest to combine all the above-mentioned bioactive heterocyclic rings attach together in a molecular framework of pyrazolone in a proper manner to enhance their biological activity. Put on all ideas in mind and continuation of earlier studies, we synthesized 4- chloro-3-methyl-5-oxo-N-phenyl-4,5-dihydro-1H-pyrazole-1-carbothioamide, 4-chloro-3- methyl-N-(2-methylphe-nyl)-5-oxo-4,5-dihydro-1H pyrazole-1-carbothioamide and N-(2- methoxyphenyl)-4-chloro -3-methyl-5-oxo-4,5-dihydro-1H pyrazole-1-carbothioamide with engrossing structural features. Moreover, the antifungal potentiality of pyrazolone and to probe the structure activity- relationship, all the fabricated molecules were evaluated for their antifungal activities in Table 1.
The synthetic pathway of the reaction, ethylacetoacetate with thiosemicarbazide to gave direct regain to the desired 4 -chloro-3-methyl-5-oxo-N-phenyl-4,5- dihydro-1H-pyrazole-1-carbothioamide, 4-chloro-3 -methyl-N-(2-methylphe-nyl)-5-oxo-4,5-dihydro-1H-pyrazole-1-carbothioamide and N-(2- methoxyphenyl)-4-chloro -3-methyl-5-oxo-4,5- dihydro-1H-pyrazole-1-carbothioamide. The initial preparation of precursors N-(substituted phenyl) thiosemicarbazide. The reactive ethylacetoacetate were approchable via the reaction of an equimolar quantity of thiosemicarbazide containing H, CH3 and OCH3 group in dimethylformamide at 80-90oC, which resulted in the formation of 4-chloro-3-methyl-5-oxo-N-phenyl-4,5-dihydro-1H-pyrazole-1-carbothioamide, 4-chloro-3-methyl-N-(2-methylphenyl)-5-oxo-4,5-dihydro-1H-pyrazole-1-carbothioamide and N-(2- methoxyphenyl)-4-chloro -3-methyl- 5-oxo-4,5-dihydro-1H-pyra-zole-1-carbothioamide in good yield.
General Procedure for the Synthesis of Pyrazolones: [3a-g]
Equimolar mixture of phenyl thiosemicarbazide (0.01 mole, 1.67 gm) (1) and ethyl- 2-chloro acetoacetate (0.01 mole, 1. 28 mL) (2) were refluxed for 10 h in DMF (20 mL). Now mixture was refrigerated with water and solidified filtrate were recrystallized with ethanol.
4-chloro-3-methyl-5-oxo-N-phenyl-4,5-dihydro-1H-pyrazole-1-carbothioamide (3a)
Yield-70%, M.P.-145oC, IR(KBr, νmax cm-1): 3330-3000(N-H), 1215(C=S), 3020-1731(C-H), 1483-1402(C-N), 755 (C-Cl); 1HNMR=(400MHz, CDCl3)δ=1.32(3H, s, CH3), 2.26(1H, s, NH ), 3.49(1H, s, C-H), 7.22-7.67(Ar-5H, m)ppm; 13C NMR: 169.13, 145.33, 137.81, 129.71, 126.09, 124.19, 77.31, 61.44, 44.40, 38.12, 24.56; MS(EI) m/z=267.02[M]+,Anal. calcd. for C11H10ClN3OS: C,49.35; H,3.76; N,15.69; Found: C,49.75; H, 3.94; N, 15.97.
4-chloro-3-methyl-N-(2-methylphenyl)-5-oxo-4,5-dihydro-1H-pyrazole-1-carbothioamide (3b)
Yield-67%, M.P.-225oC, IR(KBr, νmax cm-1): 3178(N-H-Str), 1699 (C=O), 1452 (C-N-Str), 1157 (C=S), 1602 (C=N), 755 (C-Cl); 1HNMR=(400MHz, CDCl3)δ=1.32(3H, s, CH3), 2.26(1H, s, NH ), 3.59(3H, s, Ar-CH3), 7.22-7.67(Ar-5H, m)ppm; 13C NMR: 169.13, 145.33, 137.81, 129.71, 126.09, 124.19, 77.31, 61.44, 62.8, 44.40, 38.12, 24.56; MS(EI) m/z=281.04[M]+ , Anal. calcd. for C12H12ClN3OS: C,51.15; H,4.29; N,14.91; Found: C,51.33; H, 4.94; N, 14.99.
4-chloro-3-methyl-N-(3-methylphenyl)-5-oxo-4,5-dihydro-1H-pyrazole-1-carbothioamide (3c)
Yield-68%, M.P.-179oC, IR(KBr, νmax cm-1): 3265 (N-H-Str), 1672 (C=O), 1476 (C-N-Str), 1166 (C=S), 1620 (C=N), 755 (C-Cl); 1HNMR=(400MHz, CDCl3)δ=1.32(3H, s, CH3), 2.26(1H, s, NH ), 3.59(3H, s, Ar-CH3), 7.22-7.67(Ar-5H, m)ppm; 13C NMR: 169.13, 145.33, 137.81, 129.71, 126.09, 124.19, 77.31, 61.44, 62.8, 44.40, 38.12, 24.56; MS(EI) m/z=281.04[M]+ , Anal. calcd. for C12H12ClN3OS: C,51.15; H,4.29; N,14.91; Found: C,51.34; H, 4.64; N, 14.97.
4-chloro-3-methyl-N-(4-methylphenyl)-5-oxo-4,5-dihydro-1H-pyrazole-1-carbothioamide (3d)
Yield-60%, M.P.-190oC, IR(KBr, νmax cm-1): 3256 (N-H-Str), 1668 (C=O), 1482 (C-N-Str), 1141 (C=S), 1605 (C=N), 755 (C-Cl); 1HNMR=(400MHz, CDCl3)δ=1.32(3H, s, CH3), 2.26(1H, s, NH ), 3.59(3H, s, Ar-CH3), 7.22-7.67(Ar-5H, m)ppm; 13C NMR: 169.13, 145.33, 137.81, 129.71, 126.09, 124.19, 77.31, 61.44, 62.8, 44.40, 38.12, 24.56; MS(EI) m/z=281.04[M]+ , Anal. calcd. for C12H12ClN3OS: C,51.15; H,4.29; N,14.91; Found: C,51.19; H, 4.49; N, 14.99.
N-(2-methoxyphenyl)-4-chloro-3-methyl-5-oxo-4,5-dihydro-1H-pyrazole-1-carbothioamide (3e)
Yield-64%, M.P.-235oC, IR(KBr, νmax cm-1): 3300 (N-H-Str), 1686 (C=O), 1463 (C-N-Str), 1163 (C=S), 1615 (C=N), 755 (C-Cl); 1HNMR=(400MHz, CDCl3)δ=1.32(3H, s, CH3), 2.26(1H, s, NH), 3.49(3H, s, Ar-OCH3), 7.22-7.67(Ar-5H, m)ppm; 13C NMR: 169.13, 145.33, 137.81, 129.71, 126.09, 124.19, 77.31, 61.44, 62.8, 44.40, 38.12, 24.56; MS(EI) m/z=297.03[M]+ , Anal. calcd. for C12H12ClN3O2S: C,48.40; H,4.06; N,14.11; Found: C, 48.60; H, 4.24; N, 14.67.
N-(3-methoxyphenyl)-4-chloro-3-methyl-5-oxo-4,5-dihydro-1H-pyrazole-1-carbothioamide (3f)
Yield-58%, M.P.-245oC, IR(KBr, νmax cm-1): 3245 (N-H-Str), 1660 (C=O), 1455 (C-N-Str), 1145 (C=S), 1612 (C=N), 755 (C-Cl); 1HNMR=(400MHz, CDCl3)δ=1.32(3H, s, CH3), 2.26(1H, s, NH), 3.49(3H, s, Ar-OCH3), 7.22-7.67(Ar-5H, m)ppm; 13C NMR: 169.13, 145.33, 137.81, 129.71, 126.09, 124.19, 77.31, 61.44, 62.8, 44.40, 38.12, 24.56; MS(EI) m/z=297.03[M]+ , Anal. calcd. for C12H12ClN3O2S: C, 48.40; H, 4.06; N, 14.11; Found: C, 48.93; H, 4.44; N, 14.27.
N-(4-methoxyphenyl)-4-chloro-3-methyl-5-oxo-4,5-dihydro-1H-pyrazole-1-carbothioamide (3g)
Yield-60% , M.P.-232oC, IR(KBr, νmax cm-1): 3223 (N-H-Str), 1630 (C=O), 1435 (C-N-Str), 1159 (C=S), 1606 (C=N), 755 (C-Cl); 1HNMR=(400MHz, CDCl3)δ=1.32(3H, s, CH3), 2.26(1H, s, NH), 3.49(3H, s, Ar-OCH3), 7.22-7.67(Ar-5H, m)ppm; 13C NMR: 169.13, 145.33, 137.81, 129.71, 126.09, 124.19, 77.31, 61.44, 62.8, 44.40, 38.12, 24.56; MS(EI) m/z=297.03[M]+ , Anal. calcd. for C12H12ClN3O2S: C, 48.40; H, 4.06; N, 14.11; Found: C, 48.55; H, 4.34; N, 14.55.
Disc Diffusion Assay
It is done by using Kirby-Bauer method to determine the antibacterial susceptibility at a fixed concentration [24Bauer, A.W.; Kirby, W.M.; Sherris, J.C.; Turck, M. Antibiotic susceptibility testing by a standardized single disk method. Am. J. Clin. Pathol., 1966, 45(4), 493-496.
[PMID: 5325707] ]. For this, few colonies of organism were inoculated in 2-5 ml broth and grown for 2.5 hours. Before inoculation, dried agar plates to prevent flow of inoculated material during incubation. A sterile cotton swab is dipped into the bacterial suspension and used to evenly spread the diluted culture on the agar surface. After the inoculation dried, impregnated discs were placed on the agar surface with flamed forceps and gently pressed down to ensure contact [25Pfaller, M.A.; Burmeister, L.; Bartlett, M.S.; Rinaldi, M.G. Multicenter evaluation of four methods of yeast inoculum preparation. J. Clin. Microbiol., 1988, 26(8), 1437-1441.
[PMID: 3049651] ]. The sterile (6 mm diameter) discs impregnated with fixed doses 600µg/ml were assassible on the pre-inoculated accede. The seeded petri-dishes were incubated within 30 minutes at 37oC for 48 hours. Similar plates were prepared for the standard drug, the reference antifungal drug fluconazole was used. Dimethylformamide was used as control solvent in the assay. The zone of inhibition is directly proportional to the degree of sensitivity of fungal strain and the concentration of compound. The antifungal activity of the compounds was calculated by using the formula as given below:
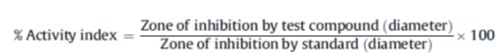
Determination of Minimum Inhibitory Concentration (MIC) Value
The antibacterial screening was determined using disc diffusion method by measuring zone of inhibition in mm.
Biological Assay
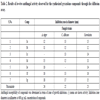
The antifungal activity of all synthesized compounds (3a-g) were tested against three fungal strains, viz; A. niger, C. albicans and Curvularia, using disc diffusion technique. Compounds (3a-g) were liquefy in dimethylformamide at different concentrations and the discs were dipped into the respective compounds and put on the petri dishes of specified organisms against a control of dimethylformamide. After 48 hours, the zone of inhibition was mascurated in mm. and the details of antifungal activity were furnished in Table 2.
RESULTS AND DISCUSSION
Synthetic route of the target compounds (3a-g) is depicted in Scheme 1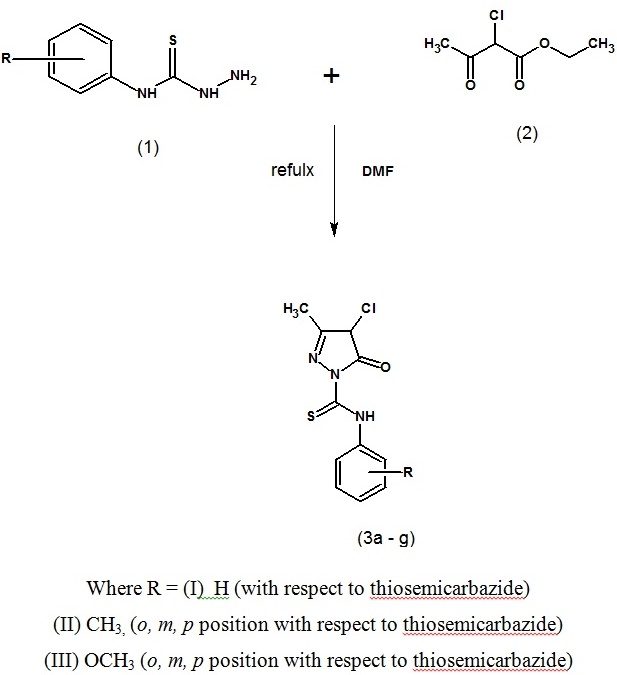 . Here, we reported newly synthesized biologically active pyrazolone derivatives by the reaction of thiosemicarbazide and ethyl-2-chloro acetoacetate with DMF as a solvent. Dimethylformamide is a polar (hydrophilic) aprotic solvent with a high boiling point as compared to ethanol. It can easily react with NHNH2SCN reactant compound and makes it possible to react with ethyl-2- choloro acetoacetate and leads to the formation of target compounds (3a-g).
. Here, we reported newly synthesized biologically active pyrazolone derivatives by the reaction of thiosemicarbazide and ethyl-2-chloro acetoacetate with DMF as a solvent. Dimethylformamide is a polar (hydrophilic) aprotic solvent with a high boiling point as compared to ethanol. It can easily react with NHNH2SCN reactant compound and makes it possible to react with ethyl-2- choloro acetoacetate and leads to the formation of target compounds (3a-g).
 |
Scheme. 1 Synthesis of substituted pyrazolone derivatives. |
The structures of the synthesized compounds (3a-g) were determined on the basis of their FT-IR, 1H-NMR, 13C-NMR and elemental analysis. The IR spectra of target compounds (3a-g) showed the presence of these groups N-H, C=O, C- N, C-Cl and C=S absorptions band at 3,200-3,430 cm-1, 1,600 cm-1, 750 cm-1, 1400-1490 cm-1 and 1,150 cm,-1 respectively. In 1H- NMR spectra, all the compounds were characterized due to the presence of aromatic protons (Ar-CH) expected multiplet near δ 7.22-7.67 ppm. Another N-H protons also exhibited singlets at δ 2.20- 2.30 ppm, respectively whereas, the pyrazolone methyl protons seemed high as a singlet near δ 1.32 ppm. 13C NMR spectra recorded signals correspond to thiosemicarbazide moiety and other aromatic corbons. The mass spectrum of pyrazolone (3a) sustained molecular ion peak at m/z= 267.02 (M)+, with the molecular formula C11H10ClN3OS. And all compounds gave satisfactory elemental analysis.
Antifungal Activity
The antifungal activities were found of compounds (3a-g) against Candida albicans, Aspergillus niger and Curvularia fungal strain. Table 2 shows result of in- vitro antifungal bioassay. The reference antifungal drug was Fluconazole. Compounds 3d and 3g were assigned to inhibit the growth of Aspergillus niger and Candida albicans, respectively, compounds 3c and 3f exhibited remarkable inhibition on Curvularia, respectively. Here we can conclude that different methyl and methoxy group on aromatic ring increases the antifungal activity of different derivatives.
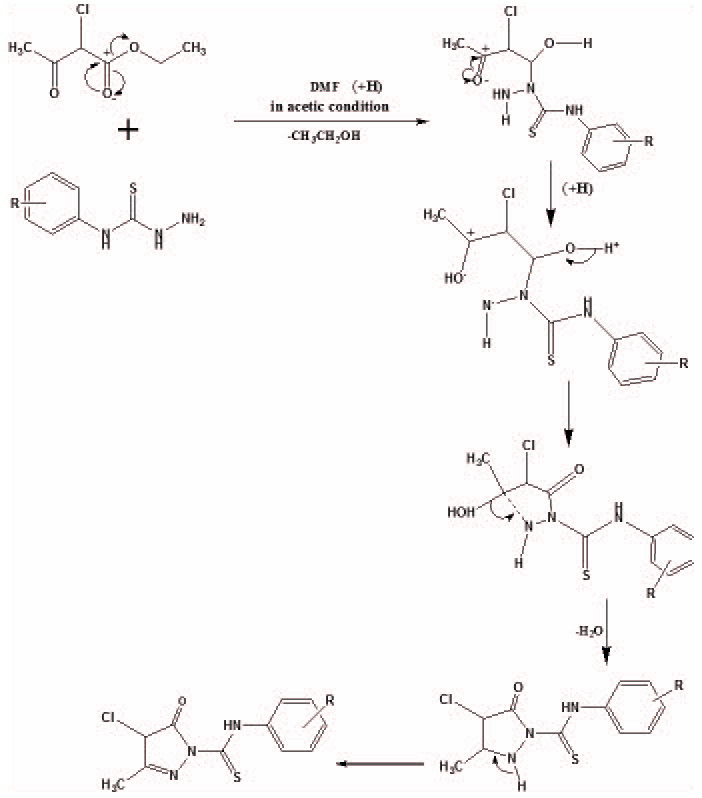 |
Proposed Mechanism of Pyrazolones Formation |
CONCLUSION
In the present work, substituted pyrazolones have been successfully synthesized by using thiosemicarbazide and ethyl-2-chloro acetoacetate as a starting materials. The synthesized compounds were deduced by spectral analysis (1H -NMR, 13C -NMR, IR, Mass) and elemental analysis. The result of screening clearly indicated the nature of substitution in newly synthesized compounds affected in-vitro antifungal activity. The presence of electron-withdrawing group on the aromatic ring of thiosemicarbazide increases the antifungal activity of tested compounds. Here, electron-donating group were also shows moderate activity against tested pathogens such as Aspergillus niger, Candida albicans and Curvularia. All the synthesized compounds exposed better antifungal activities against a wide range of microorganisms.
SUPPLEMENTARY MATERIAL
Supplementary material is available on the publishers web site along with the published article.CONFLICT OF INTEREST
The authors confirm that this article content has no conflict of interest.
ACKNOWLEDGEMENTS
We are grateful to the Head of the Department, School of studies in Chemistry, Jiwaji University Gwalior and Dean, Birla Institute of Medical Research and College of Life Sciences, Gwalior.
REFERENCES
| [1] | William, D.A.; Lemke, T.L. Principles of Medicinal Chemistry; Williams and Wilkins: Lippincott, 2002, vol. 5, p. 36. |
| [2] | Pandeya, S.N. A Text Book of Medicinal Chemistry; SG Publisher: Singapore, 2004, vol. 1, pp. 2-3. |
| [3] | Lednicer, D.; Mitscher, L.A. In Organic Chemistry of Drug Synthesis; Wiley Interscienc: New York, 1997, vol. 1, p. 226. |
| [4] | Himly, M.; Jahn-Schmid, B.; Pittertschatscher, K.; Bohle, B.; Grubmayr, K.; Ferreira, F.; Ebner, H.; Ebner, C. Ig E-mediated immediate-type hypersensitivity to the pyrazolone drug propyhenazone. J. Allergy Clin. Immunol., 2003, 111, 882-888. [http://dx.doi.org/10.1067/mai.2003.163] [PMID: 12704373] |
| [5] | Al-Haiza, M.A.; El-Assiery, S.A.; Sayed, G.H. Synthesis and potential antimicrobial activity of some new compounds containing the pyrazol-3-one moiety. Acta Pharm., 2001, 51, 251-261. |
| [6] | Castagnolo, D.; Manetti, F.; Radi, M.; Bechi, B.; Pagano, M.; De Logu, A.; Meleddu, R.; Saddi, M.; Botta, M. Synthesis, biological evaluation, and SAR study of novel pyrazole analogues as inhibitors of Mycobacterium tuberculosis: part 2. Synthesis of rigid pyrazolones. Bioorg. Med. Chem., 2009, 17(15), 5716-5721. [http://dx.doi.org/10.1016/j.bmc.2009.05.058] [PMID: 19581099] |
| [7] | Radi, M.; Bernardo, V.; Bechi, B.; Castagnolo, D. Microwave-assisted organocatalytic multicomponent Knoevenagel/hetero Diels-Alder reaction for the synthesis of 2,3-dihydropyran[2,3-c]pyrazoles. Tetrahedron Lett., 2009, 50, 6572-6575. [http://dx.doi.org/10.1016/j.tetlet.2009.09.047] |
| [8] | Moreau, F.; Desroy, N.; Genevard, J.M.; Vongsouthi, V.; Gerusz, V.; Le Fralliec, G.; Oliveira, C.; Floquet, S.; Denis, A.; Escaich, S.; Wolf, K.; Busemann, M.; Aschenbrenner, A. Discovery of new Gram-negative antivirulence drugs: structure and properties of novel E. coli WaaC inhibitors. Bioorg. Med. Chem. Lett., 2008, 18(14), 4022-4026. [http://dx.doi.org/10.1016/j.bmcl.2008.05.117] [PMID: 18571407] |
| [9] | Sauzem, P.D.; Machado, P.; Rubin, M.A.; da S Sant’anna, G.; Faber, H.B.; de Souza, A.H.; Mello, C.F.; Beck, P.; Burrow, R.A.; Bonacorso, H.G.; Zanatta, N.; Martins, M.A. Design and microwave-assisted synthesis of 5-trifluoromethyl-4,5-dihydro-1H-pyrazoles: novel agents with analgesic and anti-inflammatory properties. Eur. J. Med. Chem., 2008, 43(6), 1237-1247. [http://dx.doi.org/10.1016/j.ejmech.2007.07.018] [PMID: 17889969] |
| [10] | Pasha, F.A.; Muddassar, M.; Neaz, M.M.; Cho, S.J. Pharmacophore and docking-based combined in-silico study of KDR inhibitors. J. Mol. Graph. Model., 2009, 28(1), 54-61. [http://dx.doi.org/10.1016/j.jmgm.2009.04.006] [PMID: 19447057] |
| [11] | Rosiere, C.E.; Grossman, M.I. An analog of histamine that stimulates gastric acid secretion without other actions of histamine. Science, 1951, 113(2945), 651-653. [http://dx.doi.org/10.1126/science.113.2945.651] [PMID: 14845699] |
| [12] | Bailey, D.M.; Hansen, P.E.; Hlavac, A.G.; Baizman, E.R.; Pearl, J.; DeFelice, A.F.; Feigenson, M.E. 3,4-Diphenyl-1H-pyrazole-1-propanamine antidepressants. J. Med. Chem., 1985, 28(2), 256-260. [http://dx.doi.org/10.1021/jm00380a020] [PMID: 3968690] |
| [13] | Chauhan, P.M.; Singh, S.; Chatterjee, R.K. Antifilarial profile of substituted pyrazoles: a new class of antifilarial agents. Indian J. Chem. Sect. B, 1993, 32, 858-861. |
| [14] | Shestopalov, A.M.; Emelyanova, Y.M.; Shestopalov, A.A.; Rodinovskaya, L.A.; Niazimbetova, Z.I.; Evans, D.H. Cross of substituted 6-amino-spiro-4-(N-R-piperidine-4’)-2H,4H-pyrano[2,3-condensation of derivatives of cyanoacetic acid and carbonyl compounds. Part 1. One-stage synthesis c]pyrazole-5-carbonitriles. Tetrahedron, 2003, 59, 491-7496. [http://dx.doi.org/10.1016/S0040-4020(03)01178-5] |
| [15] | Guangfei, L.; Lang, Li.; Dianzeng, J.; Kaibei, Yu. Synthesis, structure and photochromic properties of 1-phenyl-3-methyl-4-(4-bromobenzal)-pyrazolone-5 thiosemicarbazone. Struct. Chem., 2005, 16, 135-140. [http://dx.doi.org/10.1007/s11224-005-2837-6] |
| [16] | Baciu-Atudosie, L.; Ghinet, A.; Belei, D.; Gautret, P.; Rigo, B.; Bicu, E. An efficient one-pot reaction for the synthesis of pyrazolones bearing a phenothiazine unit. Tetrahedron Lett., 2012, 53, 6127-6131. [http://dx.doi.org/10.1016/j.tetlet.2012.08.152] |
| [17] | Anonymous. British Pharmacopoeia; H. M. S. Publication Center: UK, 1988, vol. 2, p. A205. |
| [18] | Ochei, J.; Kolhatkar, A. Medicinal Laboratory Science-Theory and Pracitces; Tata McGrow-Hill Publishing Co. Ltd.: New Delhi, 2000, pp. 808-818. |
| [19] | Barry, A.L. The Antimicrobial Susceptibility Test: Principal and Practices; lllu lea and Febiger Publishers: Philadephia, Pa (U.S.A), 1976, pp. 180-195. |
| [20] | Isloor, A.M.; Kalluraya, B.; Sridhar Pai, K. Synthesis, characterization and biological activities of some new benzo[b]thiophene derivatives. Eur. J. Med. Chem., 2010, 45(2), 825-830. [http://dx.doi.org/10.1016/j.ejmech.2009.11.015] [PMID: 19945198] |
| [21] | Chandrakantha, B.; Shetty, P.; Nambiyar, V.; Isloor, N.; Isloor, A.M. Synthesis, characterization and biological activity of some new 1,3,4-oxadiazole bearing 2-flouro-4-methoxy phenyl moiety. Eur. J. Med. Chem., 2010, 45(3), 1206-1210. [http://dx.doi.org/10.1016/j.ejmech.2009.11.046] [PMID: 20004043] |
| [22] | Sankappa Rai, U.; Isloor, A.M.; shetty, P.; Vijesh, A.M.; Prabhu, N.; Isloor, S.; Thiageeswaran, M.; Fun, H.K. Novel chromeno [2,3-b]-pyrimidine derivatives as potential anti-microbial agents. Eur. J. Med. Chem., 2010, 45(6), 2695-2699. [http://dx.doi.org/10.1016/j.ejmech.2010.02.040] [PMID: 20231044] |
| [23] | Vijesh, A.M.; Isloor, A.M.; Isloor, S.K.; Shivananda, K.N.; Shyma, P.C.; Arulmoli, T. Synthesis of some new pyrazolone derivatives as potent antimicrobial agents. Der. Pharm. Chem., 2011, 3, 454-463. |
| [24] | Bauer, A.W.; Kirby, W.M.; Sherris, J.C.; Turck, M. Antibiotic susceptibility testing by a standardized single disk method. Am. J. Clin. Pathol., 1966, 45(4), 493-496. [PMID: 5325707] |
| [25] | Pfaller, M.A.; Burmeister, L.; Bartlett, M.S.; Rinaldi, M.G. Multicenter evaluation of four methods of yeast inoculum preparation. J. Clin. Microbiol., 1988, 26(8), 1437-1441. [PMID: 3049651] |