- Home
- About Journals
-
Information for Authors/ReviewersEditorial Policies
Publication Fee
Publication Cycle - Process Flowchart
Online Manuscript Submission and Tracking System
Publishing Ethics and Rectitude
Authorship
Author Benefits
Reviewer Guidelines
Guest Editor Guidelines
Peer Review Workflow
Quick Track Option
Copyediting Services
Bentham Open Membership
Bentham Open Advisory Board
Archiving Policies
Fabricating and Stating False Information
Post Publication Discussions and Corrections
Editorial Management
Advertise With Us
Funding Agencies
Rate List
Kudos
General FAQs
Special Fee Waivers and Discounts
- Contact
- Help
- About Us
- Search

The Open Biomaterials Journal
(Discontinued)
ISSN: 1876-5025 ― Volume 5, 2014
Production of Chitin and Chitosan from Partially Autolyzed Shrimp Shell Materials
Nguyen Van Toan*
Abstract
The effect of partial autolysis during storage of shrimp biowaste on the quality of the extracted chitin and chitosan has been investigated. Shrimp waste materials were stored in a 30°C incubator for 1-4 days. The quality of the chitin and chitosan extracted from these materials was assessed by ash and protein analysis and in solution by viscosimetry. Chitosan from fresh biowaste, treated for 1 day at 30°C showed the highest viscosity 3590 cps. Chitosan from frozen material showed the highest viscosity at 3580 cps after 2 days at 30°C. The corresponding values for chitosan without 30°C treatment were 2300 and 2700 cps. Normally, viscosity, solubility and molecular weight of chitosan are closely related. The data of this study show that after partial autolysis the viscosity of the chitosan obtained has increased, but that other characteristics of chitosan such as degree of deacetylation, molecular weight, solubility and turbidity did not change.
Article Information
Identifiers and Pagination:
Year: 2009Volume: 1
First Page: 21
Last Page: 24
Publisher Id: TOBIOMTJ-1-21
DOI: 10.2174/1876502500901010021
Article History:
Received Date: 17/11/2008Revision Received Date: 18/8/2009
Acceptance Date: 22/8/2009
Electronic publication date: 16/10/2009
Collection year: 2009
open-access license: This is an open access article licensed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted, non-commercial use, distribution and reproduction in any medium, provided the work is properly cited.
* Address correspondence to this author at the School of Biotechnology, International University, Vietnam National University, Ho Chi Minh City; Fax: + 84 -83-7244271 ; Email: nvtoan@hcmiu.edu.vn
| Open Peer Review Details | |||
|---|---|---|---|
| Manuscript submitted on 17-11-2008 |
Original Manuscript | Production of Chitin and Chitosan from Partially Autolyzed Shrimp Shell Materials | |
INTRODUCTION
The crude shrimp head and skin materials have only a low economical value and are treated as biowaste or sold to animal feed manufacturers [1]. The shrimp biowaste in the tropical region contains 10-20 % calcium, 30-65% protein content and 8- 10% chitin on a dry basis [2]. Chitosan, which is obtained by N- deacetylation of chitin, has attracted attention due to its unique cationic character. Typically, chitosan is produced from waste generated from crustacean processing (e.g. shrimp and crab). Chitin and its derivative chitosan are of commercial interest due to their excellent biocompatibility, biodegradability, non-toxicity, chelating and adsorption power. With these characteristics, especially chitosan has many attractive applications in biotechnology, food and pharmaceutical industry, in cosmetics, environmental engineering, in agriculture and aquaculture. Though chemical procedures are widely used to produce chitin and chitosan utilizing shrimp waste as raw material [3], there is not much information how to improve the production process so as to produce chitosan with consistent quality, especially from stored material sources. The purpose of this study is to include partial autolysis in the existing protocol in order to improve the quality of chitosan product. This paper describes experiments to determine a suitable process for chitin/chitosan production from partially autolysed shrimp shell and suggests utilizing shrimp shells that have softened and partially degraded under carefully defined conditions.
MATERIALS AND METHODS
Shrimp shells from black tiger shrimp (Penaeus monodon) were provided by a local shrimp processing factory. The shrimp shells were collected in big plastic bags with addition of ice to preserve the shrimp shell from decomposition. During transportation to the laboratory (transport time was only one and a half hour), the shrimp waste was kept cool and in the dark to avoid effects of direct sunlight. After arrival, the shrimp waste was directly used to produce chitosan as specified below. In this paper shrimp shell without further treatment is named fresh shrimp shell, shrimp shell stored in the freezer (at minus 18°C) for several months is named frozen shrimp shell.
Various procedures have been adopted to obtain high quality chitin by removing protein, inorganic material (mainly CaCO3) and pigments and lipid. Demineralization is usually carried out by treatment with HCl and deproteination by treatment with NaOH but other researchers have used other methods as well. The sequence of these processes has been varied, although in most chitin producing industries deproteination is carried out prior to demineralization [4]. In this research a modified method has been used in which shrimp shell was allowed to undergo partial autolysis before it was used to produce chitin and chitosan. The selected autolysis times were 1, 2, 3 and 4 days. Fresh shrimp shell used as the control for this experiment is marked as 0 day treatment (Fig. 1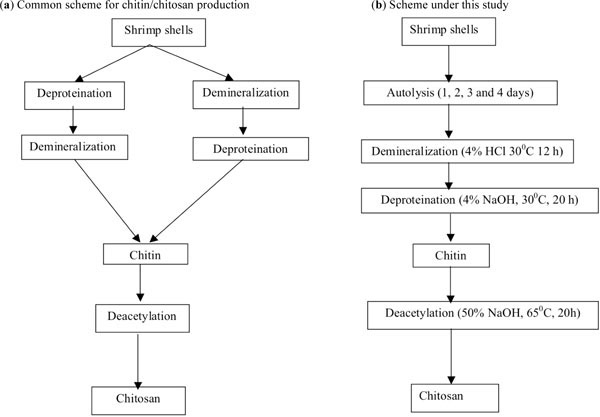 ). The autolyzed shrimp shell was demineralized with 0.68 M HCl solution (1:5 w/v) at ambient temperature (28-32°C) for 12 h. The residue was washed and soaked in tap water for 6-8 h. It was then dewatered and deproteinized with 0.62 M NaOH solution (1:5 w/v) at ambient temperature for 20 h.
). The autolyzed shrimp shell was demineralized with 0.68 M HCl solution (1:5 w/v) at ambient temperature (28-32°C) for 12 h. The residue was washed and soaked in tap water for 6-8 h. It was then dewatered and deproteinized with 0.62 M NaOH solution (1:5 w/v) at ambient temperature for 20 h.
 |
Fig. (1) A comparison between (a) the common process and (b) revised process under this study for the production of chitin and chitosan. |
The chitin obtained from the revised process above was deacetylated in 12.5 M NaOH (1:5 w/v) solution at 65°C for 20 h. After deacetylation, the chitosan was washed and dried in sunlight and assayed for moisture content, ash content, protein content, degree of deacetylation, viscosity, solubility, turbidity and molecular weight [5]. The protein content in the chitosan sample was determined using the micro-biuret method [6]. The degree of deacetylation was analyzed by first derivative ultraviolet (UV) spectrophotometry [7]. Weight average molecular weight was determined by gel permeation chromatography (Waters GPC) with a differential refractometer detector. Dextrans of various molecular weights ranging from 9.9 x 103 to 2 x 106 Kd were used as standards. Ash content was determined by the standard AOAC method [8]. Various physico-chemical criteria were investigated under standard conditions, for chitosan in a 1% solution in 0.35 M acetic acid. Turbidity was assessed using a turbidity meter (Model 2100P portable turbidity meter, HACH Company, USA) and viscosity by a Brookfield Model DV - VII + Viscometer. Solubility was measured using the transglucosidase method [9]: the pH of 50 ml 1% (w/v) chitosan solution was adjusted to 4.8 with 30% (w/v) sodium acetate. Transglucosidase L-500 (Genencor International, 500 µl) was added. After incubation at 60°C for 24 h, the insoluble material was collected by filtration using a pre-weighed Whatmann GF/C filter paper (1.2 µm). The filter paper was dried and weighed and the amount of insoluble material was calculated from its weight gain.
RESULTS AND DISCUSSION
Characterization of Chitosan Samples
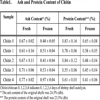
The deproteinized and demineralized shrimp shell, chitin, was assayed for ash and protein content to check if the two critical values are below the level of 1%. For chitosan, product properties such as the degree of deacetylation, solubility and turbidity were assessed to characterize the preparations. Table 1 shows the ash and protein content in the chitin samples.
Chitosan samples obtained from the two different production schemes were characterized and compared with each other. All chitosan samples had a uniform quality with less than 1% protein content (by microbiuret assay), less then 1% ash and about 8-10% moisture content. Most samples had a degree of deacetylation close to 90%, solubility near 100% and turbidity 20 ~ 22 NTU. Data on solubility, turbidity, degree of deacetylation and molecular weight indicate that there are no significant differences in these parameters among all treatments for the chitosan produced from fresh and frozen shrimp shell. There are, however, differences in the ash content, protein content and in particular, in the viscosity of chitosan solutions (Tables 2 and 3).
The protein content of chitosan samples in all treatments are well below 1% (Tables 2 and 3). The protein content ranges from 0.53% to 0.94% and 0.38% to 0.66% for the fresh and frozen shrimp shells, respectively. It can be seen that the protein content of chitosan produced from fresh shrimp shells is higher than that of frozen shrimp shells in every treatment. This may be because of the matrix of protein and another component in the shrimp shell is softened by the freezing procedure. The frozen shell might have a more open structure in which the matrixes of proteins, inorganic materials and lipids have already been weakened during the storage time. Hence, it allows NaOH solution to easily breakdown the protein and to remove it from the shell. The same phenomenon has been observed when using HCl solution to demineralize inorganic materials from shrimp shells.
The Phenomenon of Improved Chitosan Quality After Partial Autolysis
Limited autolysis of shrimp biomaterial has a positive effect on the quality of the extracted chitin and chitosan. The data presented in this paper show that demineralization becomes somewhat more efficient, protein removal is facilitated and the viscosity of the chitosan is significantly enhanced.
Regarding demineralization, it was also observed that after partial autolysis mineral removal can be achieved under more mild conditions. Normally, the demineralization process is conducted in 1 M HCl for a period of 12 h. As shown for partial autolyzed material in Table 4, even 0.5M HCl can reduce the ash content to less than 1% within 6h of treatment.
Demineralization might be improved by partial autolysis due to a direct action on CaCO3 or by an action on the protein scaffold, resulting in an improved exposure of the CaCO3. For the industrial practice of chitin extraction this means that a shorter time and lower concentration of HCl may suffice and that damage due to hydrolytic action of HCl on the polymer backbone structure of chitin can be reduced.
Following partial autolysis, deproteination is more efficient as well. The protein content after treatment of autolyzed biomaterial with 2% NaOH is similar to the protein content after standard 4% NaOH treatment. More surprisingly, it took not more than 6h to obtain a residual protein level of 0.31%. Apparently partial autolysis changes the chemical structure of the shell tissue, resulting in a higher sensitivity for the alkali mediated peptide hydrolysis. Since low concentration of alkali is equally effective, the cost of deproteination in industry and the environmental impact of this treatment will be reduced.
Partial autolysis also results in a chitosan product with a higher viscosity. This has been observed for fresh chitosan kept during 1 day at 30°C degrees as for chitosan from frozen material, stored 2 days at 30°C degrees. This could be caused by a variety of factors. The processing procedure has been investigated in detail to reveal variables that might explain this peculiar phenomenon. The most realistic explanation is that as result of autolysis, all steps in the chemical treatment of the biowaste are facilitated allowing for the extraction of a chitosan with a minimum of chemical damage. As a result the chitosan will be isolated in its best form and generate upon dissolution a high viscosity.
CONCLUSIONS
The viscosity of a chitosan sample is a crucial parameter that indicates the quality of the chitosan sample. From the results obtained through this study, it can be concluded that chitosan samples with an enhanced viscosity can be obtained when the shrimp biowaste is allowed to a limited decay. In particular, the viscosity of chitosan samples extracted from fresh shells and treated for 1 day and from frozen shells treated for 2 days show a remarkable higher viscosity.
Normally, viscosity, solubility and molecular weight of chitosan are closely related. But the result of this study shows that in spite of the increase of the viscosity, other characteristics of chitosan such as the degree of deacetylation, molecular weight, solubility and turbidity have not changed compared to the chitosan from fresh material.
ACKNOWLEDGEMENT
The author would like to express his thanks to the International University, Vietnam National University, Ho Chi Minh City for generous financial support.