- Home
- About Journals
-
Information for Authors/ReviewersEditorial Policies
Publication Fee
Publication Cycle - Process Flowchart
Online Manuscript Submission and Tracking System
Publishing Ethics and Rectitude
Authorship
Author Benefits
Reviewer Guidelines
Guest Editor Guidelines
Peer Review Workflow
Quick Track Option
Copyediting Services
Bentham Open Membership
Bentham Open Advisory Board
Archiving Policies
Fabricating and Stating False Information
Post Publication Discussions and Corrections
Editorial Management
Advertise With Us
Funding Agencies
Rate List
Kudos
General FAQs
Special Fee Waivers and Discounts
- Contact
- Help
- About Us
- Search

The Open Biomaterials Journal
(Discontinued)
ISSN: 1876-5025 ― Volume 5, 2014
Improved Chitin and Chitosan Production from Black Tiger Shrimp Shells Using Salicylic Acid Pretreatment
Nguyen Van Toan*
Abstract
Shrimp chitin and chitosan with improved characteristics were isolated from black tiger shrimp waste pretreated with 0.04 M Salicylic acid for ten hours. The pretreated shrimp shells could be efficiently demineralized and deproteinized at ambient temperature using 0.680 M HCl and 0.620 M NaOH, respectively. The duration of the treatments was 8 hours, the ash and protein residues in the final chitosan were about 0.48% and 0.51% respectively; the viscosity was 4800 cps; the solubility was up to 98%. In comparison with treatment at ambient temperature (30oC) without pretreatment, the chemical consumption, the duration of the treatment, ash and protein residues was reduced to 75-20%, whereas viscosity and absence of insolubles improved by a factor of 2.5.
Article Information
Identifiers and Pagination:
Year: 2011Volume: 3
First Page: 1
Last Page: 3
Publisher Id: TOBIOMTJ-3-1
DOI: 10.2174/1876502501103010001
Article History:
Received Date: 12/9/2009Revision Received Date: 11/11/2010
Acceptance Date: 16/12/2010
Electronic publication date: 31/1/2011
Collection year: 2011
open-access license: This is an open access article licensed under the terms of the Creative Commons Attribution Non-Commercial License ("http://creativecommons.org/licenses/by-nc/3.0/"), which permits unrestricted, non-commercial use, distribution and reproduction in any medium, provided the work is properly cited.
* Address correspondence to this author at the School of Biotechnology, International University, Ho Chi Minh City, Vietnam; Fax: +84-83-7244271; E-mail: nvtoan@hcmiu.edu.vn
| Open Peer Review Details | |||
|---|---|---|---|
| Manuscript submitted on 12-9-2009 |
Original Manuscript | Improved Chitin and Chitosan Production from Black Tiger Shrimp Shells Using Salicylic Acid Pretreatment | |
INTRODUCTION
Chitin and chitosan are the structural components in the cuticles of crustacean, insects, mollusk and in the cell wall of fungi and plant pathogens (Suwalee, 2002) [1]. Reportedly, chitin and chitosan have various biological functions, for instance, antimicrobial activity; wound healing, especially on the proliferative phase and matrix formation.
Since the early eighties, chitin and its derivatives have been used for various biomedical purposes especially in skin and wound management products in Japan [2]. Though several standard chemical procedures have been widely used to produce chitin and chitosan utilizing shrimp shell waste as raw material, there is not much information about how to improve the production process so as to produce chitosan with consistent quality especially high solubility, high viscosity and high stability in degree of deacetylation while ash content ash and protein are as low as possible.
Salicylic acid is a keratolytic agent that softens the hyperkeratotic areas by dissolving the intra-cellular matrix and enhancing shedding of scales. This acid has been widely used in the removal of all types of human and animal skin warts using a technique of cauterization wherein slaked lime is applied to wart and then the surface of wart is scratched by using the stem of betel leaf, in general [3]. It has been shown that Salicylic acid softens keratin, loosens cornified epithelium and causes swelling even of viable cells. In a test of percutaneous absorption through skin of hairless rat as a good example, the data showed that Salicylic acid softens the horny layer and enhances the permeability of the stratum corneum.
Exoskeletal chitin-protein complexes give protection against chitinases and mild alkaline conditions. The more strongly the protein is bound to chitin, the harder for NaOH to penetrate and to detach the protein from the solid shell material. Salicylic acid seems to weaken the exoskeletal complex matrix of protein resulting in the removal of a protein fraction that otherwise remains bound to the solid waste. As a consequence, this acid helps allowing NaOH solution to easily penetrate and facilitate the breakdown of protein components in the matrix.
This paper describes experiments to determine the suitable process for production of improved characteristics of chitin and chitosan from black tiger shrimp shells for various aspects of chitosan application.
MATERIALS AND METHODS
The black tiger shrimp shells used in this study were obtained from a local shrimp processing factory, Southern part of Vietnam. The shells which were pretreated with 0.04M Salicylic acid was demineralized with 0.680 M HCl solution (1:5 w/v) at ambient temperature, 28-32°C for 8 hrs. The residue was washed and soaked in tap water for 6-8 hours. It was then dewatered and deproteinized with 0.620 M NaOH solution (1:5 w/v) at ambient temperature for 16hrs.
The chitin obtained from the above processes was deacetylated in 12.5 M NaOH (1:5 w/v) solution at 65°C for 18 hrs for chitosan isolation.
The process scheme for chitin/chitosan production under this study is shown in Fig.(1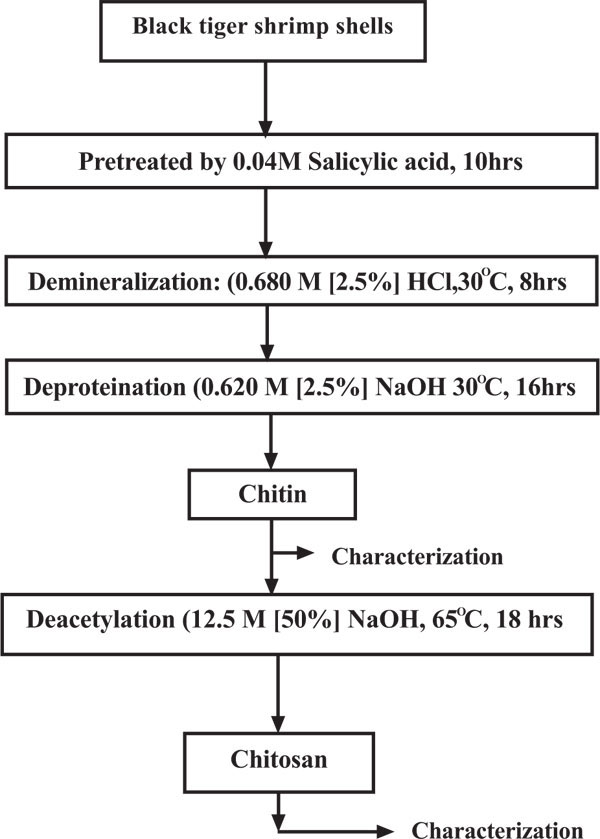 ). After deacetylation, the chitosan was washed and dried in sunlight and assayed for moisture content, ash content, protein content, degree of deacetylation, viscosity, solubility, turbidity and molecular weight.
). After deacetylation, the chitosan was washed and dried in sunlight and assayed for moisture content, ash content, protein content, degree of deacetylation, viscosity, solubility, turbidity and molecular weight.
 |
Fig. (1) The revised process for the production of chitin and chitosan under this study. |
The protein content in the chitosan sample was determined using the micro-biuret method [4]. The degree of deacetylation was determined by first derivative ultraviolet (UV) spectrophotometry [5]. Weight average molecular weight was determined by gel permeation chromatography (Waters GPC) with a differential refractometer detector [6]. Dextrans of various molecular weights ranging from 9.9 x 103 to 2 x 106 were used as standards. Ash content was determined by the standard AOAC method [7]. Various physico-chemical criteria were investigated for chitosan in a 1% solution in 0.35 M acetic acid. Turbidity was assessed using a turbidimeter (Model 2100P, HACH Company, USA) and viscosity by a Brookfield Model DV - VII + Viscometer. Solubility was measured using the transglucosidase method [8]: the pH of 50 ml 1% (w/v) chitosan solution was adjusted to 4.8 with 30% (w/v) sodium acetate and mixed with Transglucosidase L-500 (Genencor International, 500 μl). After incubating at 60°C for 24 hrs, the insoluble material was collected by filtration using a pre-weighed Whatman GF/C filter paper (1.2 μm). The filter paper was dried and weighed and the amount of insolubles was calculated from its weight gain. Turbidity was assessed using a turbidimeter (Model 2100P, HACH Company, USA) and viscosity by a Brookfield Model DV-VII+ viscometer.
The reproducibility of chitosan was determined by storing chitosan in 0.1 M acetic acid for a period of 60 days at temperature of 28°C- 30°C according to method of Stevens [9]. In this study, the molecular weight was measured every ten days.
Chitosan membrane was tested for its thickness using a Mitutoyo micrometer. Tensile strength and elongation at break were measured for sample pieces of 10 ± 1 µm thickness (10 x15 mm) with a tensile tester, model Synergie 200, Adamel Lhmargy Co. Moisture sorption of chitosan membrane was measured at 84% relative humidity (Nunthanid et al., 2001) [10]. Gel swelling index was determined by the method reported by Chen et al. (1994) [11]. Permeation of sodium salicylate through chitosan membranes was assessed using a Franz diffusion cell (5 cm effective area of diffusion) at 30 ± 2°C. The solute concentration in the receptor compartment was calculated from its absorbance at 296 nm using a spectrophotometer. Permeation coefficient was calculated by applying Fick_s law. Lysozyme activity was assayed by the amount of reducing sugar generated by incubation of chitosan membrane (1 cm2) or chitosan powder (0.05 g) in a solution of 1.25 mg lysozyme in 25 ml 0.2 M phosphate buffer pH 7. The mixture was incubated at 37°C. The reducing sugar was determined after 24 h of incubation by the Schales method (Imoto and Yagishita, 1971) [12].
All data are expressed as means ± standard deviation of representative of similar test carried out in triplicate. Statistical differences in cell number were determined by student’s t-test in which, p<0.005 was considered statistical significant.
RESULTS AND DISCUSSION
The general characteristics of chitin and chitosan produced from the pretreated shrimp shells by Salicylic acid are listed in Table 1.
The viscosity of the Salicylic acid pretreated chitosan in solution is closed to 5000 cps compared with 2650 cps of control chitosan produced using common methods.
Data on the depolymerization of chitosan, of which, was storing in 0.1 M acetic acid for a period of 60 days at a temperature of 28°C- 30°C has been compared with studies of our colleagues in Bioprocess Program laboratory in Bangkok on comparison of storage of chitosan in acetic acid and in formic acid [9]. As expected, the produced chitosan using the proposed procedures under this study showed a reduction of less than 85% in Molecular weight (MW) in the 60th day. Data on the rate of chitosan degradation in 0.1 M acetic acid is shown in the Table 2.
The produced chitosan under the revised procedure was used for preparation of chitosan membrane and testing its characteristics, of which tensile strength, moisture sorption as well as the permeability constant are especially explored. As shown in Fig. (2 ) and Table 3, the chitosan membranes were transparent and colorless. The average thickness of the membranes was 10 ± 1µm.
) and Table 3, the chitosan membranes were transparent and colorless. The average thickness of the membranes was 10 ± 1µm.
 |
Fig. (2) Appearance of chitosan membranes before and after detaching (A) and drying in the lab (B) at ambient temperature |
Data of the current research are similar to those of Stevens [9] and Trung TS [6], provided that some characteristics of chitosan such as degree of deacetylation, solubility, turbidity and viscosity are essentially important parameters and should be taken into account before starting making chitosan membrane for further applications.
CONCLUSIONS
The collected data demonstrate that the high and consistent quality of chitosan produced from black tiger shrimp shells could be obtained by pre-treating the shells using dilute Salicylic acid. These results are similar to those, of which the shrimp shells materials were preconditioned using dilute Benzoic [13]. Shrimp chitosan produced in this way could be used as a good source of raw material for further post processes of chitosan amongst other applications.
ACKNOWLEDGEMENT
The author would like to express his thanks to the International University, Vietnam National University, Ho Chi Minh City for the generous financial support.



