- Home
- About Journals
-
Information for Authors/ReviewersEditorial Policies
Publication Fee
Publication Cycle - Process Flowchart
Online Manuscript Submission and Tracking System
Publishing Ethics and Rectitude
Authorship
Author Benefits
Reviewer Guidelines
Guest Editor Guidelines
Peer Review Workflow
Quick Track Option
Copyediting Services
Bentham Open Membership
Bentham Open Advisory Board
Archiving Policies
Fabricating and Stating False Information
Post Publication Discussions and Corrections
Editorial Management
Advertise With Us
Funding Agencies
Rate List
Kudos
General FAQs
Special Fee Waivers and Discounts
- Contact
- Help
- About Us
- Search

The Open Process Chemistry Journal
(Discontinued)
ISSN: 1875-1806 ― Volume 6, 2014
Fibrillar Templating of Hydrotalcites
Alicia E. Sommer1, Pedro Bosch2, Geolar Fetter1, *
Abstract
Layered double hydroxides also known as hydrotalcites have been synthesized in presence of fibrillar organic materials: human hair, cotton or nylon. These templates differ on their diameter, hydrophobicity and surface roughness. These parameters are correlated to the morphology and texture of the resulting hydrotalcites. A rapid synthesis method based on microwave irradiation is used and the materials were characterized by complementary physical techniques.
Article Information
Identifiers and Pagination:
Year: 2009Volume: 2
First Page: 6
Last Page: 11
Publisher Id: TOCPCJ-2-6
DOI: 10.2174/1875180601002010006
Article History:
Received Date: 6/12/2008Revision Received Date: 21/1/2009
Acceptance Date: 20/2/2009
Electronic publication date: 24/3/2009
Collection year: 2009
open-access license: This is an open access article licensed under the terms of the Creative Commons Attribution Non-Commercial License(http://creativecommons.org/licenses/by-nc/3.0/), which permits unrestrictive use, distribution, and reproduction in any medium, provided the original work is properly cited.
* Address correspondence to this author at the Universidad Autónoma de Puebla, Facultad de Ciencias Químicas, Blvd. 14 Sur y Av. San Claudio, C.P. 72570 Puebla, PUE, Mexico; Fax: (52)2222443106; E-mail: geolarfetter@yahoo.com.mx
| Open Peer Review Details | |||
|---|---|---|---|
| Manuscript submitted on 6-12-2008 |
Original Manuscript | Fibrillar Templating of Hydrotalcites | |
INTRODUCTION
Layered double hydroxides (LDH’s), hydrotalcite-like compounds or hydrotalcites are anionic clays as they have positively charged layers, due to a partial substitution of a divalent metal by a trivalent one. They may be used in catalysis, adsorption or ion exchange, among others [1-3]. These materials can be represented by the general formula [M2+1-xM3+x(OH)2(Am-)x/m.nH2O where M2+ and M3+ are di- and trivalent metal cations, respectively, and Am- is a compensating anion intercalated between layers [4,5].Although it is possible to obtain various LDH’s modifying the composition and the synthesis process, the most naturally occurring LDH is Mg6Al2(OH)16CO3.4H2O. The usual synthesis procedure is co-precipitation of the cationic metals in a basic medium, followed by a hydrothermal treatment [6,7]. Some authors have reported recently on the use of templates to modify morphology and hence texture [8,9]. Those inducing agents are gas or alcohol molecules; in this case a vesicular shape is obtained. However, no other than globular or layered morphology has been reported. The particle shape is determinant if species as DNA [10], pigments [11] or polymers [12] have to be incorporated into a clay; a layered compound with a globular shape should not behave in the same way as a cylindrical one. Such features are crucial if hydrotalcites should be used as drug deliverers [3].
In this work we report on the effect of human hair, nylon and cotton used as templates in the hydrotalcite synthesis, in presence of microwave irradiation. Microwave irradiation, indeed, propitiates a rapid crystallization, a small crystallite size and the required equipment is rather simple [13-17]. The aim is to determine the effect of various cylindrical templates (hair, nylon or cotton) on the particle hydrotalcite morphology. The templates were chosen as a function of their size as well as their surface features.
EXPERIMENTAL METHODS
Hydrotalcite Synthesis
Mg/Al-hydrotalcite samples were synthesized from a Mg(NO3)2.6H2O and Al(NO3)3.9H2O (both from Aldrich) water solution (1 M), a 0.5 M NaOH (Baker) aqueous solution and three different templates. Organic templates were human hair, cotton or nylon fibers.
Over a Becker recipient containing 2 g of template and enough NaOH solution to reach a pH value of 8 (even if this is not the usual value to synthesize hydrotalcites), the Mg-Al solution was added, dropwise. The pH value was maintained constant (pH=8) adding the required amount of NaOH solution, as at higher pH values the templates decompose and agglomerate, and at lower pH values, the hydrotalcite does not precipitate. The resulting solution amounts correspond to a molar ratio Mg/Al of 3. The solution was continuously stirred.
After precipitation, the resulting gels were hydrothermally treated in a microwave autoclave (MIC-I, Sistemas y Equipos de Vidrio S.A. de C.V.) for 10 minutes at 80 ºC, operating at 2.45 GHz and a power of 200 W. The solids were recovered by decantation and washed several times with distilled water. The solids were, then, dried in an oven at 70 ºC overnight.
The samples were labeled H-MW, C-MW and N-MW depending on the used template: hair, cotton or nylon, respectively. Lastly, a reference sample, R-MW, was prepared in the same way, without template and under microwave irradiation.
Characterization
X-Ray Diffraction
A Bruker-axs D8-advance diffractometer coupled to a copper anode X-ray tube was used to identify the compounds present in the powdered samples. A diffracted beam monochromator selected the Kα radiation.
FTIR Spectroscopy
FTIR spectra in the region 4000-400 cm-1 were obtained with a Magna-IR Spectrometer 550 Nicolet. The pellets were prepared with KBr.
Nitrogen Adsorption
The BET surface areas were determined from the nitrogen adsorption-desorption curves by the conventional multipoint technique with a Micromeritics ASAP 2020. The pore size distribution curves were obtained with the BJH method applied to the desorption branch. The samples were pretreated at 200 °C for 10 hours at high vacuum.
Scanning Electron Microscopy
A scanning electron microscope LEICA, Stereoscan 440 was used. The samples were previously covered with gold to avoid charge problems.
RESULTS
X-Ray Diffraction
Fig. (1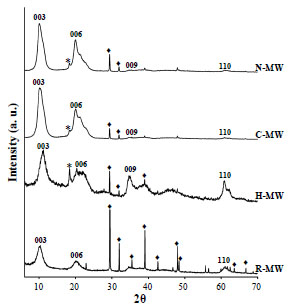 ) compares the X-ray diffraction patterns of the synthesized compounds. The R-MW sample diffractogram presents some broad and some sharp and intense peaks. The first ones correspond to hydrotalcite (JCPDS card 22700) whereas the second to nitratine (JCPDS card 361474). The nitratine precipitation is induced by the low OH concentration as pH value is 8.
) compares the X-ray diffraction patterns of the synthesized compounds. The R-MW sample diffractogram presents some broad and some sharp and intense peaks. The first ones correspond to hydrotalcite (JCPDS card 22700) whereas the second to nitratine (JCPDS card 361474). The nitratine precipitation is induced by the low OH concentration as pH value is 8.
In presence of hair, the obtained compound is again hydrotalcite; the amount of nitratine is now much lower. A third compound appears identified as gibbsite, Al(OH)3, JCPDS card 3318. If the fibers are cotton or nylon, again hydrotalcite is obtained with very small amounts of nitratine and gibbsite. In these two diffraction patterns, only the peaks 003 and 006 are evident. The 110 and 113 peaks have faded showing that order in the (a x b) plane is low, thus the arrangement of Mg and Al octahedra is not regular. Furthermore, as only the 003 and 006 peaks are present and not the 009 neither the 015, the stacking order should be very short.
The interlayer distance obtained from the 003 peak position turned out to be 8.7 Å in all samples. Thus, the compensating anions are nitrates as expected.
Infrared Spectroscopy
In all samples, the band at 3450 cm-1 is attributed to the OH structural groups and the band at 1657 cm-1 to water hydroxyls in hydrotalcites.1 The intense band at 1387 cm-1 is due to interlayered nitrates [18,19]. Hence, the infrared spectra, Fig. (2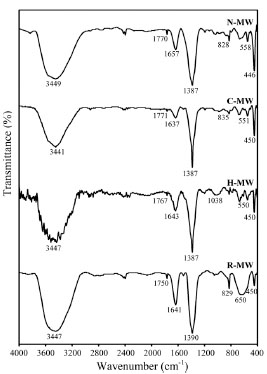 ), present the typical profile of a double layered hydroxide intercalated with nitrates. Note that in H-MW and N-MWsamples, bands at 1800 and 1500 cm-1 are not present, these bands are attributed to ammides and cheratin, therefore no residues of hair or nylon remain.
), present the typical profile of a double layered hydroxide intercalated with nitrates. Note that in H-MW and N-MWsamples, bands at 1800 and 1500 cm-1 are not present, these bands are attributed to ammides and cheratin, therefore no residues of hair or nylon remain.
Scanning Electron Microscopy
H-MW Sample Micrograph
The hair cuticle originates from a single-layered epithelium and it is made up of a large number of overlapping scales of keratin. The separation between scales is 5 to 10 μm. The epicuticle mainly made of fatty acid chains is a semi-permeable membrane which surrounds each cuticle cell and forms the outer surface of hair [20]. Human hair diameter depends on the person age and it oscillates between 90 and 70 μm.
The H-MW surface image obtained by SEM, Fig. (3 ), is homogeneous. It presents a series of ordered parallel sheets separated by a distance of 2 to 4 μm. They are densely stacked. On top, irregular particles ca. 2 μm are dispersed all over, they probably correspond to the impurities already identified by X-ray diffraction.
), is homogeneous. It presents a series of ordered parallel sheets separated by a distance of 2 to 4 μm. They are densely stacked. On top, irregular particles ca. 2 μm are dispersed all over, they probably correspond to the impurities already identified by X-ray diffraction.
 |
Fig. (3) Scanning electron micrograph of the H-MW sample (synthesis in presence of hair). a) x5000; b) x10000; c) x20000. |
C-MW Sample Micrograph
Cotton fiber is covered by a thin membrane or cuticle. The cotton fiber diameter is ca. 10-20 μm. Cotton fiber presents several key properties such as hydrophobicity, good affinity for hydrocarbons, rapid adsorption on contact, and high adsorption and retention through interfibre capillaries. The difference between cotton and hair is that cotton is a much thinner fiber (four times less) and that the surface is smooth. The corresponding micrograph, Fig. (4 ), shows a material composed by very large chunks (10 to 20 μm) which in turn are constituted by packed flakes, 1 to 5 μm whose thickness is less than 0.1 μm.
), shows a material composed by very large chunks (10 to 20 μm) which in turn are constituted by packed flakes, 1 to 5 μm whose thickness is less than 0.1 μm.
 |
Fig. (4) Scanning electron micrograph of the hydrotalcite prepared in presence of cotton, C-MW. a) x5000; b) x10000; c) x20000. |
N-MW Sample Micrograph
Nylon fibers are artificial polyamide polymers which are obtained from a diacid polycondensed with a diamine. The surface is smooth and presents more hydrophobic features than cotton. The fiber diameter is in this case close to 15 μm. In this sample, again, large globular particles are found (ca. 4 μm, Fig. 5 ). The hydrotalcite flakes are stacked in a more disordered way as the layers cross each other.
). The hydrotalcite flakes are stacked in a more disordered way as the layers cross each other.
 |
Fig. (5) Scanning electron micrograph of the sample prepared in presence of nylon, N-MW.a) x5000; b) x10000; c) x20000. |
Nytrogen Physisorption
Fig. (6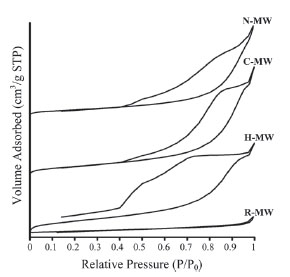 ) compares the adsorption-desorption cycles of R-MW, H-MW, C-MW and N-MW samples. The curves are type IV and the hysteresis loops type B, following the IUPAC classification. Hence, pores are open at both extremes. The reference hydrotalcite, sample R-MW, differs from the three other materials. In this preparation the pore size distribution is narrow whereas in the other samples the distribution is very broad, mainly in the H-MW. The corresponding pore size distributions are shown in Fig. (7
) compares the adsorption-desorption cycles of R-MW, H-MW, C-MW and N-MW samples. The curves are type IV and the hysteresis loops type B, following the IUPAC classification. Hence, pores are open at both extremes. The reference hydrotalcite, sample R-MW, differs from the three other materials. In this preparation the pore size distribution is narrow whereas in the other samples the distribution is very broad, mainly in the H-MW. The corresponding pore size distributions are shown in Fig. (7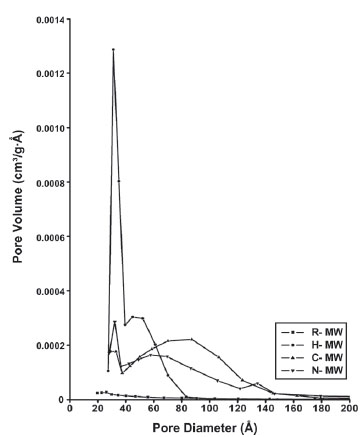 ). The R-MW sample, whose surface area is 1 m2/g, does not show any micropores. Hair, which is the larger template, generates a material whose pore size distribution is monomodal with a maximum at 30 Å; the surface area of this material is 8 m2/g. The C-MW and N-MW preparations, whose surface areas are 5 m2/g, present broad similar pore size distributions, from 20 to 150 Å with maxima, in C-MW at 100 Å and, in N-MW at 70 Å. It seems, then, that the template size, as well as the hydrophobicity, determines the pore size distributions as hair is much larger (70-90 μm) than nylon or cotton (10-20 μm) which only differ in their hydrophobicity.
). The R-MW sample, whose surface area is 1 m2/g, does not show any micropores. Hair, which is the larger template, generates a material whose pore size distribution is monomodal with a maximum at 30 Å; the surface area of this material is 8 m2/g. The C-MW and N-MW preparations, whose surface areas are 5 m2/g, present broad similar pore size distributions, from 20 to 150 Å with maxima, in C-MW at 100 Å and, in N-MW at 70 Å. It seems, then, that the template size, as well as the hydrophobicity, determines the pore size distributions as hair is much larger (70-90 μm) than nylon or cotton (10-20 μm) which only differ in their hydrophobicity.
 |
Fig. (6) Nitrogen adsorption-desorption isotherms of the samples. |
 |
Fig. (7) Pore size distributions of the samples obtained by the BJH method. |
DISCUSSION
Three hydrotalcites have been prepared with similar structure as shown by X-ray diffraction, but with different textural and morphological features depending on the size and hydrophobicity of the fibrous template. They were all prepared at a pH value of 8, which ensures the integrity of the fibrillar inductors. Use of hair as a template is not new, as it has been used to prepare mesoporous silica tubes [21]. The authors report that not only a tubular morphology is obtained at a micron scale but also a nanometrical mesophase. The assembly of silica tetrahedra is very easily accomplished as the angle between tetrahedra may cover a broad range, from 180 to 60°. Instead, the magnesium or aluminum octahedra of hydrotalcite tend to form rigid layers.
In our sample, only the mesophase is obtained. In the micrographs, a longitudinal order of the hydrotalcite particles is observed. Hair is a low hydrophobic material whose surface may be fully covered by water molecules which, in turn, promote a high concentration of hydrotalcite precursors. The hair scale borders are probably nucleation centers, as they are covered by acid chains, which, in a basic medium, become negatively charged. The magnesium and aluminum cations are thus attracted, following parallel lines to the scale border. Indeed, they are separated ca. 4 μm, which is the distance between scales. It seems then that rings of hydrotalcite are formed all around the hair template. Microwave irradiation favors the formation of a large number of hydrotalcite seeds all around the scale border.
Such hypothesis is consistent with the results obtained with C-MW and N-MW samples, which do not present such ordered morphology. These two fibers have a lower diameter and therefore the surface is more curved. The adhered Mg,Al octahedra, thus, cannot adhere to such a curved surface. Furthermore, there are no nucleation centers as these templates have no scales. Precursors, then, form the rigid layers observed in both samples. Such is not the case if alumina is synthesized, as biomorphic Al2O3 fibers have been prepared using natural fibers of cotton as bio-templates [22].
A second mechanism could be the one proposed by Carmody et al. [23]. Cotton fiber has at its surface a complex mixture of molecules made from carbon and hydrogen with a significant proportion of non-polar groups present. These non-polar groups have low surface energy and would be attracted to the oil molecules, but would repel our hydrotalcite aqueous precursors. Therefore, this should cause the formation of droplets in our sample. With temperature, small independent hydrotalcite particles are formed which are weakly bonded to the fiber. The use of microwave irradiation, of course, favors the formation of very small and hot droplets which result in well structured hydrotalcite small particles.
These trends are confirmed by the specific surface area values. Hair provides smaller particles and therefore a higher specific surface area. Only in this sample, pores close to 30 Å diameter are determined; this porosity is probably due to the assemblage of the hydrotalcite particles forming rings around hair. Instead, in the other two materials, specific surface areas are smaller, as hydrotalcite particles are large and well structured. The corresponding pore size distributions follow the same trend, only large pores are observed and they are due to interparticle spaces. The small values of specific surface areas are due to the well known blocking role of nitrates, as discussed in previous publications [24-26]. The differences in gibbsite amount in each sample can be correlated to the differences in OH surface content of the three templates. The hydrotalcite contains more aluminum in the low containing gibbsite samples.
CONCLUSION
The comparison among the hydrotalcites prepared using various organic fibrillar templates (hair, cotton or nylon) has shown that the interactions between the template and the hydrotalcite precursors determine the hydrotalcite morphology and texture. The hydrotalcite particle association forming rings around the fibrillar template or flakes weakly retained to cotton or nylon depends on hydrophobicity, as well as on roughness and surface curvature. Last but not least, microwave irradiation preparation technique is a simple and fast method to obtain these materials. It guarantees a high number of nucleation centers.
ACKNOWLEDGMENT
The authors acknowledge the financial support of CONACYT. The technical work in X-ray diffraction and electron microscopy of Leticia Baños and Omar Novelo is gratefully recognized.


