- Home
- About Journals
-
Information for Authors/ReviewersEditorial Policies
Publication Fee
Publication Cycle - Process Flowchart
Online Manuscript Submission and Tracking System
Publishing Ethics and Rectitude
Authorship
Author Benefits
Reviewer Guidelines
Guest Editor Guidelines
Peer Review Workflow
Quick Track Option
Copyediting Services
Bentham Open Membership
Bentham Open Advisory Board
Archiving Policies
Fabricating and Stating False Information
Post Publication Discussions and Corrections
Editorial Management
Advertise With Us
Funding Agencies
Rate List
Kudos
General FAQs
Special Fee Waivers and Discounts
- Contact
- Help
- About Us
- Search

The Open Environmental Research Journal
Formerly: The Open Ecology Journal
ISSN: 2590-2776 ― Volume 16, 2023
- New Journal Website
- Journal Home
- Editorial Board
- Board Recruitment Workflow
- Instructions for Authors
- Plagiarism Prevention
- Fabricating and Stating False Information
- Research Misconduct
- Post Publication Discussions and Corrections
- Allegations from Whistleblowers
- Advertising Policy
- Short Guide
- Editorial Policies
- Publishing Ethics and Rectitude
- Quick Track Option
- Reviewer Guidelines
- Guidelines for Guest Editors
- Publication Fee
- Publication Cycle - Process Flowchart
- Archiving Policies
- Special Issues
A Quantitative Assessment of Germination Parameters: the Case of Crotalaria Persica and Tephrosia Apollinea
Fatima Al-Ansari, Taoufik Ksiksi*
Abstract
The present investigation attempted to assess the impact of seed pre-treatments on the germination of Crotalaria persica (Burm. f.) Merr and Tephrosia apollinea (L) Pers. Equally important was our investigation to quantitatively compare different germination parameters used for seed germination studies. The results showed that the final germination was highest for both species under chipping (CHIP), sand paper (SP) and 20 to 90 minutes in concentrated sulfuric acid (SA20 to SA90). Rubbing in sand (SAND) and water-soaking (SOAK) showed lower germination percentages. Mean germination time (MGT) was highest after 50 minutes in sulfuric acid (SA50) for both C. persica and T. apollinea. The coefficient of velocity of germination (CVG) was highest when seeds were treated with sulfuric acid during 30 minutes (SA30) for C. persica (56.1%) and during 60 minutes (SA60) for T. apollinea (66.8%). For the germination index (GI), chipping and all SA exposures (except 90 minutes) revealed high means for both C. persica and T. apollinea, while SP also resulted in high germination index for C. persica. The mean rate of seed germination was highest under sand rubbing and distilled water (0.9 and 0.8, respectively) for C. persica and under SA20 (1.3), for T. apollinea. The germination rate index (GRI) for C. persica was highest under CHIP, SP, and SA20 to SA60. Timson Germination Index (TGI) showed high values for most pre-treatments.
The highest TGI recorded was 45.6% for SA30. As for the correlation between the germination parameters, it is recommended that final germination percent coupled with germination index are used as germination parameters. They do not require complex calculations, while they complement each other in giving a representative evaluation of seed germination.
Article Information
Identifiers and Pagination:
Year: 2016Volume: 9
First Page: 13
Last Page: 21
Publisher Id: TOECOLJ-9-1-13
DOI: 10.2174/1874213001609010013
Article History:
Received Date: 16/08/2015Revision Received Date: 16/08/2016
Acceptance Date: 30/08/2016
Electronic publication date: 17/11/2016
Collection year: 2016
open-access license: This is an open access article licensed under the terms of the Creative Commons Attribution-Non-Commercial 4.0 International Public License (CC BY-NC 4.0) (https://creativecommons.org/licenses/by-nc/4.0/legalcode), which permits unrestricted, non-commercial use, distribution and reproduction in any medium, provided the work is properly cited.
* Address correspondence to this author at the Biology Department, UAE University, PO Box 15551 Al-Ain, UAE; Tel: +97137136548; Fax: +97137134927; E-mail: tksiksi@uaeu.ac.ae
| Open Peer Review Details | |||
|---|---|---|---|
| Manuscript submitted on 16-08-2015 |
Original Manuscript | A Quantitative Assessment of Germination Parameters: the Case of Crotalaria Persica and Tephrosia Apollinea | |
1. INTRODUCTION
One of the most important processes in the plant natural life cycles is seed germination. For any seeds to initiate the process of germination, they have to imbibe water under favorable environmental conditions (Tobe et al., 2001). The process is affected by several factors such as temperature variations, oxygen levels, water availability and lack of inhibitory environmental conditions. However, unfavorable conditions in some species could lead to seed dormancy (Yildiztugay & Kucukoduk, 2012). Dormancy was classified into five classes, with physiological dormancy being the most extensive scheme (Baskin & Baskin, 2004). Seed dormancy and germination are complex features relating to higher plants which are influenced by factors that relate to environmental conditions (Koornneef et al., 2002). Seed coat dormancy or physical dormancy play an important role in the success of the germination process. Seed coat dormancy is caused by a hard and impermeable seed coat that prevents imbibitions and some-times gaseous exchange (Yildiztugay & Kucukoduk, 2012). Physical dormancy (Baskin & Baskin, 2004) differs between species type, degrees of drought, and the stage of maturation. This type of dormancy can be pre-treated through the use of mechanical resistance or physical impermeability, which depends on the thickness of the seed coat (Schmidt, 2000). One widely used chemical to break seed dormancy is the use of sulfuric acid. Effectiveness of the pre-treatment improved as the time of exposure to sulfuric acid increased (Wang et al., 2007).
Moreover, time, rate, homogeneity, and synchronization of germination are also critical issues (Ranal & Santana, 2006) simply because the dynamics of the germination process are indicative of the ecology of any plant species. These indicators and features are useful for physiologists, seed technologists and ecologists (Ranal & Santana, 2006). The ecology of any plant species population could be better predicted using appropriate germination indices, to successfully establish and survive as seed germination involves qualitative responses of individuals as well as population responses which are distributed over time (Scott 30 et al., 1984).
Many procedures for data analysis of seed germination responses are scattered throughout the literature. Many, including Timson germination index, have been reported as a good indicator of seed germination rate (Al-Mudaris, 1998). This index is calculated as the sum of percent germination of seeds at 2 day intervals by the total germination period (Khan & Ungar, 1996). Timson’s index is widely used and praised (Brown & Mayer, 1988; Ansari et al., 2012; Dwiyanti et al., 2014) while setbacks to Timson method have also been reported (Al-Mudaris, 1998). He reported with varying examples that different Timson germination indices could still be reached, adding levels of complications and interpretations (Al-Mudaris, 1998). Similar critisism was reported by Heydecker (1966). He reported that Timson’s method would be as successful an attempt as any simpler ones. Heydecker (1966) also suggests that Timson’s index may lead to obscurity, except when germination rates are both rapid and high or both slow and low. Brown & Mayer (1988) studied many germination indices and coefficients to evaluate their appropriateness in distinguishing high and low germination. All, except Timson’s germination index method, were no better for ranking germination responses than total germination (Brown & Mayer, 1988). Many of the reported methods are normally relating germination percentage to some form of time taken for the germination rate to be achieved (Timson, 1965). This simplistic approach generally leads to difficulties in comparing data from different trials and in assessing germination velocity (Timson, 1965). More complicated indices and coefficients have also been suggested. The generalized Richards function, with a procedure of parallel curve analysis, to assess 55 germinations was reported (Berry et al., 1988). This method also calculates a number of derived quantities including maximum daily rate of germination and time to 50% of final germination (Berry et al., 1988). Additionally, Al-Mudaris (1998) assessed eight different germination parameters including some standard ones (e.g. first day of germination) and others more complicated such as the co-efficient of velocity of germination. Ranal et al. (2009) also reported detailed calculation steps relating to mean germination time and mean germination rate.
Here an attempt is made to assess some of the existing methods relating to germination indices, to compare the information they provide and to correlate them against the most widely used index (ie. Timson’s). Comparisons are made between Timson’s germination index vs. six other methods: final germination percentage, coefficient of velocity of germination, germination rate index and germination index as reported by Al-Mudaris (1998) and mean germination time and mean germination rate as reported by Ranal et al. (2009). A discussion of the most appropriate seed pre-treatment for superior germination rates is also included. To accomplish the aims of the present study, we use germination trial data of Crotalaria persica and Tephrosia apollinea. The two species are of important ecological benefits in the desert systems of the UAE.
2. MATERIALS AND METHODS
2.1. Plant Species
Crotalaria persica (Burm. f.) Merr, known locally as Nazaa, is a perennial leafless shrub, which is uncommon in the UAE found mostly in Ras Al-Kaimah and Al Ain. It is also found in some habitats such as sandy desert dunes and gravel flats. This plant is flowering from January to March (Karim & Fawzi, 2007). Crotalaria persica is considered as a forage species and serves as a medicinal plant (Mosti et al., 2012).
Tephrosia apollinea (L) Pers, locally known as hailara, is a perennial, woody and leafy species found in the UAE. Stems much-branched, erect, up to 70 cm. Bush can be up to 100 cm across. It is one of the most common plants in the lower mountains throughout the UAE. It is not grazed and tend to be a dominant plant and is widely considered as an indicator of overgrazing (Jongbloed et al., 2003).
2.2. Seed Germination and Pre-treatments
Seeds from the two species C. persica and T. apollinea were collected from Al-Ain city - UAE (N24◦ 200 - E55◦ 750). The weather in Al-Ain is hot during the summer and cool in winter. It is distinguished with dry and low rainfall area.
Each pre-treatment was performed in a separate experiment with a total of 20 seeds for each pre-treatment/replicate combination, placed in 9 cm petri dish on one layer of filter paper and covered with a second layer. Each pre-treatment was replicated four times. The seeds were supplied daily with distilled water during the germination trial which lasted fifteen days. All petri dishes were kept in an incubator at 25oC with continuous light. The germination recordings were carried out every morning at 9 am and the germinated seeds were picked out from the petri dishes after counting. At the 8th day, all remaining seeds were washed with 4% bleach and the filter papers were changed to avoid bacterial growth. The pre-treatments tested were: manual chipping across the seed embryo (CHIP), rinsing with distilled water (DW) at an average temperature of about 80oC as a control, rubbing the seed with sand for 2-3 minutes (SAND), rubbing the seeds with sand paper for few minutes (SP), soaking in water for 24 hours (SOAK), and exposed to sulfuric acid (96% H2SO4) during 20, 30, 40, 50, 60 and 90 minutes (SA20, SA30, SA40, SA50, SA60, and SA90, respectively).
2.3. Methods of Calculations of Germination Parameters
Seven different germination parameters were assessed. The methodology of calculations of parameters 1, 2, 4 and 5 followed Al-Mudaris (1998), 3 and 6 followed Ranal et al. (2009) while Timson’s germination index was calculated using the formula reported by Ajmal Khan & Ungar (1998).
- Final germination percentage (G) = the total seeds germinated at end of trial/number of initial seeds used 100 times.
- Mean germination time (MGT) = ΣFx/ΣF; where F is the number of seeds germinated on day x.
- Mean germination rate (MGR) = CV/100 = 1/T; where T is mean germination time and CV: coefficient of velocity.
- Coefficient of velocity of germination (CVG) = N1+N2+...+Ni/100 x N1T1+...+NiTi;
- where N is the number of seeds germinated every day and T is the number of days from seeding corresponding to N.
- Germination rate index (GRI) = G1/1+G2/2+....+Gi/i; where G1 is the germination percentage on day 1, G2 is the germination perentage at day 2; and so on.
- Germination index (GI) = (20xN1)+(19xN2)+ ... +(1xN20); where N1, N2 ...N20 is the number of germinated seeds on the first, second and subsequent days until 20th day and the multipliers (e.g. 20, 19 ...etc.) are weights given to the days of the germination.
- Timson germination index (TGI) = ΣG/T, where G is the percentage of seed germinated per day, and T is the germination period.
2.4. Statistical Analyses
SPSS (Arbuckle, 2010) was used to perform a descriptive analysis based on means of each germination index. One-way ANOVA was used to compare means of appropriate experimental treatments. Pair-wise correlations were also carried out using SPSS (Arbuckle, 2010). As Timson’s germination index (TGI) has been widely used (Rincker, 1983; Pammenter et al., 1984; Brown & Mayer, 1988; Ranal & Santana, 2006), the correlations were assessed between TGI against each of the other six indices: G, CVG, GRI, GI, MGT and MGR.
3. RESULTS
3.1. Seed Pre-treatments Effects on Final Germination
Fig. (1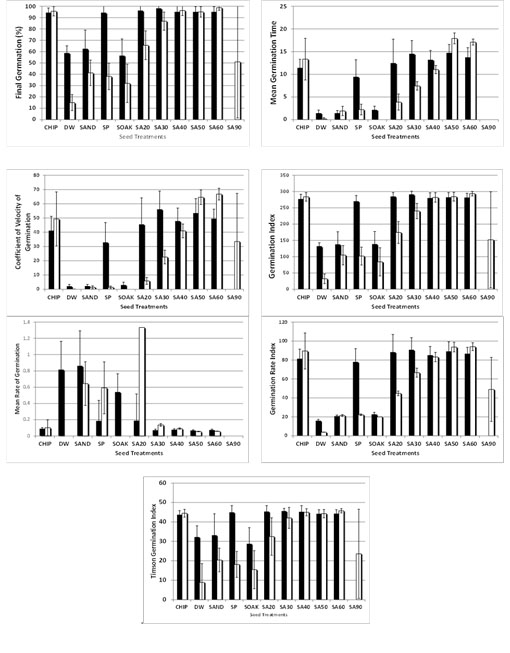 ) shows mean variations for all seven germination indices as affected by the seed pre-treatment (ie. chipping, distilled water, sanding, sand paper, soaking and the six levels of uric acid exposures).
) shows mean variations for all seven germination indices as affected by the seed pre-treatment (ie. chipping, distilled water, sanding, sand paper, soaking and the six levels of uric acid exposures).
The final germination was highest for C. persica seeds under chipping, sand paper and sulfuric acid exposures (except SA90) treatments. Seeds treated with SA90 did not germinate. For T. apollinea, chipping resulted in a 95.6% final germination and SA40 resulted in a 96.2% germination. Soaking in distilled water did not work well with an average germination of 15%. The mean germination time (MGT) was lowest under distilled water for C. persica and T. apollinea. SA90 was excluded because of ratio errors; none of the seeds germinated. The average coefficient of velocity of germination was highest when treated with SA30 and SA60 for C. persica and T. apollinea, respectively (56.1% and 66.8%). When using the germination index, chipping and all SA exposures (except SA90) revealed high means for both C. persica and T. apollinea. Sand paper resulted in high germination index for C. persica. The mean rate of germination was highest under sand paper and distilled water pre-treatments (0.9 and 0.5; respectively) for C. persica and under SA20, with an average of 1.3, for T. apollinea. The germination rate index (GRI) for C. persica was highest under chipping, sand paper, and all SA exposures (except for SA90). For T. apollinea, chipping also resulted in GRI, but it is important to highlight the increasing GRI means from low SA exposure (SA20) to higher exposures, up until SA60. The results for Timson Germination Index (TGI) show high values for all treatments; except for SA90.
3.2. Correlations Analyses of Germination Indices
As described in the methodology section, the correlation analyses were conducted between TGI and every other six indices under investigation. The aim was to assess the agreement of each index with the most commonly used germination index (i.e. TGI).
For C. persica, all six indices significantly correlated with TGI at P<0.001 (Fig. 2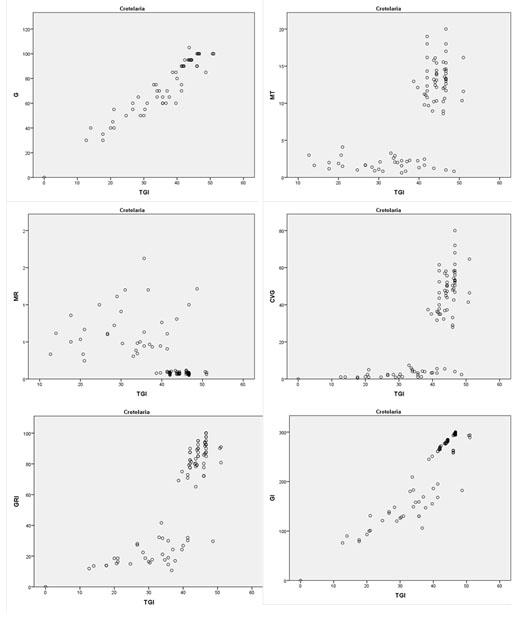 , Table 1). But the highest Pearson correlation was observed between TGI and the final germination (G) at P<0.001 (Table 1). The correlation between TGI and germination index (GI) revealed a Pearson correlation of 0.91 (P<0.001). The lowest correlation was between TGI and the mean germination rate (MR) with an average of -0.52 at P<0.001 (Table 1).
, Table 1). But the highest Pearson correlation was observed between TGI and the final germination (G) at P<0.001 (Table 1). The correlation between TGI and germination index (GI) revealed a Pearson correlation of 0.91 (P<0.001). The lowest correlation was between TGI and the mean germination rate (MR) with an average of -0.52 at P<0.001 (Table 1).
For T. apollinea, all indices had significant correlations with TGI, except for the mean rate of germination (MR) with a Pearson coefficient of -0.4 at P=0.33 (Fig. 3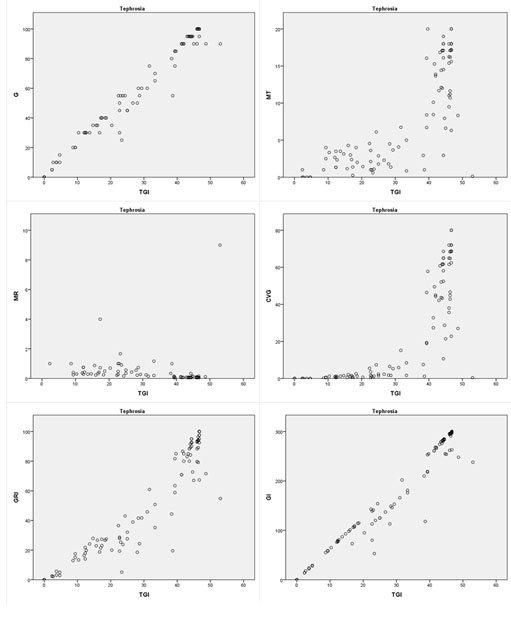 , Table 1). The highest Pearson correlation was between TGI and final germination (G) at P<0.001. Germination index was the second highly correlated with TGI with a Pearson coefficient of 0.97 at P<0.001 (Table 1).
, Table 1). The highest Pearson correlation was between TGI and final germination (G) at P<0.001. Germination index was the second highly correlated with TGI with a Pearson coefficient of 0.97 at P<0.001 (Table 1).
4. DISCUSSION
Here, an attempt was made to assess the correlations between Timson germination index (TGI), a widely used germination index, against six other germination parameters. The most appropriate seed pre-treatment options to break the dormancy for both C. persica and T. apollinea were also discussed. The overall aim was to recommend germination parameters that require less mathematical sophistication with reliable outcomes. Simply because too many procedures for data analysis of seed germination responses are reported by Scott et al. (1984).
The final germination was highest for C. persica seeds under chipping, sand paper and all, except one, sulfuric acid exposures that were tested. Manual chipping of Accacia spp. seeds provided larger improvements in germination rate, 190 and the seeds began to germinate faster than under hot water (Clemens et al., 1977). While a chipping pretreatment did not give as good a germination percentage as other pretreatments for Alstroemeria seeds (King & Bridgen, 1990).
The average coefficient of velocity of germination was highest when treated with SA30 and SA60 for C. persica and T. apollinea, respectively (56.1% and 66.8%).
The highest germination was observed in Tamarindus indica seeds treated with 50% Sulfuric acid exposure (Muhammad & Amusa, 2004). Timson Germination Index (TGI) also showed high values for all treatments; except for SA90. High acid exposures may have been the most important factor leading to this lack of germination. Low germination rates (50% vs. 28.6%) were also reported for Parkia biglobosa (Aliero, 2004). He added that prolonged exposures to SA may be detrimental to seeds as the acid may damage vital parts of the embryo. All species (other than Cassia obtusifolia), including T. purpurea, had strong integumental inhibition which was easily eliminated by mechanical scarification or immersion in concentrated Sulfuric acid (Sy et al., 2001). Crotalaria ochroleuca had emergence rates above 15% compared with <10% for T. radicans (Tauro et al., 2009).
It is recommended that final germination percent coupled with germination index are used as germination parameters. First, both parameters require limited calculation steps. Second, both parameters provide information on germination levels as well as temporal aspects of germination. Third, they both provided the highest correlation with TGI using seeds from C. persica and T. apollinea. For C. persica, all six indices significantly correlated with TGI. But the highest Pearson correlation was observed between TGI and the final germination (G) and between TGI and germination index (GI). These parameters were successfully used for sunflower hybrid seeds (Ahmad, 2001). Germination rate index and the germination index provided reliable information. Germination rate index gave the highest correlation between seed characteristics and seedling germination (Ahmad, 2001). Moreover, using combined indices avoids reliance on single parameter that may provide misleading information. As 220 several single-value germination indices, were tested with limited success (Brown & Mayer, 1988) it was also recommended that germination data are not always accompanied by some information on the time taken for the stated percentage to be reached (Timson, 1965). As discussed earlier, the most widely used TGI has its drawbacks. Different germination percentages, using TGI, could still attain the same index, thus complicating interpretations (Al-Mudaris, 1998). More complicated parameters and calculations may also provide reliable germination information (Berry et al., 1988), but it is more practical to use simpler indices.
CONFLICT OF INTEREST
The authors confirm that this article content has no conflict of interest.
ACKNOWLEDGEMENTS
A sincere thank you to our colleagues at the Biology Department and the UAEU for providing an environment that encourages collaboration and research. Partial funds were received from NWC-31R044 to pay for publication fees.








