- Home
- About Journals
-
Information for Authors/ReviewersEditorial Policies
Publication Fee
Publication Cycle - Process Flowchart
Online Manuscript Submission and Tracking System
Publishing Ethics and Rectitude
Authorship
Author Benefits
Reviewer Guidelines
Guest Editor Guidelines
Peer Review Workflow
Quick Track Option
Copyediting Services
Bentham Open Membership
Bentham Open Advisory Board
Archiving Policies
Fabricating and Stating False Information
Post Publication Discussions and Corrections
Editorial Management
Advertise With Us
Funding Agencies
Rate List
Kudos
General FAQs
Special Fee Waivers and Discounts
- Contact
- Help
- About Us
- Search

The Open Virology Journal
(Discontinued)
ISSN: 1874-3579 ― Volume 15, 2021
The Quest for Materials-Based Hydrogels with Antimicrobial and Antiviral Potentialities Alternate Title: Materials-Based Hydrogels Antimicrobial and Antiviral
Hafiz M. N. Iqbal*
Abstract
In recent years, the Antimicrobial Resistance (AMR) or Multidrug Resistance (MDR) and viral infections have become serious health issues, globally. Finally, after decades of negligence, the AMR/MDR and viral infection issues have now captured a worldwide attention of the global leaders, public health community, legalization authorities, academia, research-based organizations, and medicinal sector of the modern world, alike. Aiming to resolve these issues, various methodological approaches have been exploited, in the past several years. Among them, biomaterials-based therapeutic hydrogels are of supreme interests for an enhanced and efficient delivery in the current biomedical sector. Depending on the regulatory authorities and practices, the antibiotics consumption was expedited than ever before driven by rising and increasing access, across the globe. Though the emergence of AMR/MDR in microorganisms and emergence/reemergence of viral infections are considered as a natural phenomenon, however, these concerning issues have been driven by those mentioned above faulty human behavior. In this context, many scientists, around the globe, are working at wider spectrum to resolve this problematic issue, efficiently. A proper understanding of biological mechanisms is essential to combat this global threat to the living beings. In this review, an effort has been made to highlight the potent features of materials based hydrogels possessing antimicrobial and antiviral potentialities. The information is also given on the potential research activities, and possible mechanisms of actions of hydrogels are discussed with a closeup look at the future recommendations.
Article Information
Identifiers and Pagination:
Year: 2018Volume: 12
Issue: Suppl-2, M4
First Page: 69
Last Page: 79
Publisher Id: TOVJ-12-69
DOI: 10.2174/1874357901812010069
Article History:
Received Date: 17/5/2017Revision Received Date: 14/3/2018
Acceptance Date: 14/5/2018
Electronic publication date: 31/08/2018
Collection year: 2018
open-access license: This is an open access article distributed under the terms of the Creative Commons Attribution 4.0 International Public License (CC-BY 4.0), a copy of which is available at: (https://creativecommons.org/licenses/by/4.0/legalcode). This license permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
* Address correspondence to this author at the Tecnologico de Monterrey, School of Engineering and Sciences, Campus Monterrey, Ave. Eugenio Garza Sada 2501, Monterrey, N.L., CP 64849, Mexico; Tel: +52 81 8358 2000; E-mails: hafiz.iqbal@my.westminster.ac.uk, hafiz.iqbal@itesm.mx
| Open Peer Review Details | |||
|---|---|---|---|
| Manuscript submitted on 17-5-2017 |
Original Manuscript | The Quest for Materials-Based Hydrogels with Antimicrobial and Antiviral Potentialities | |
1. INTRODUCTION
Materials-based hydrogels are a three-dimensional cross-linked polymeric network which can be multifunctional in nature and able to respond to the external stimuli e.g. environmental pH, temperature, light, electric field, etc. Most often, the biomaterials e.g. chitosan, etc. based hydrogels possess excellent biocompatibility features, thus are broadly practiced in various sectors of pharmaceutical, cosmeceutical and biomedical engineering [1Berger J, Reist M, Mayer JM, Felt O, Peppas NA, Gurny R. Structure and interactions in covalently and ionically crosslinked chitosan hydrogels for biomedical applications. Eur J Pharm Biopharm 2004; 57(1): 19-34.
[http://dx.doi.org/10.1016/S0939-6411(03)00161-9] [PMID: 14729078] -4Iqbal HMN. Biomaterials: A trendy source to engineer functional entities-An overview. arXiv preprint 2018; arXiv:1803.05972. Available from:
https://arxiv.org/abs/1803.05972]. Also, owing to the water holding capacity, hydrogels have an extraordinary capability to swell and/or de-swell depending on the environmental conditions accordingly. The available hydrophilic groups or functional domains i.e. carboxyl, amino, and/or hydroxyl groups in the polymeric network play a major role in the water holding capacity of the hydrogels [5Pal K, Banthia AK, Majumdar DK. Polymeric hydrogels: Characterization and biomedical applications. Des Monomers Polym 2009; 12(3): 197-220.
[http://dx.doi.org/10.1163/156855509X436030] ]. The hydrophilic groups or functional domains and water holding feature of a hydrogel are directly proportional to each other. Higher the hydrophilic groups or functional domains, higher will be the swelling or water holding capacity. The swelling behavior can be controlled by mimicking the cross-linking density of a polymeric network. During water exposure, water penetrates into the polymeric networks causing swelling, thus responsible for giving hydrogel its shape. Likewise, a living tissue, a hydrogel in its complete swollen state has some unique characteristics such as soft and rubbery consistency along with a low interfacial tension with water or biological fluids [6Hamidi M, Azadi A, Rafiei P. Hydrogel nanoparticles in drug delivery. Adv Drug Deliv Rev 2008; 60(15): 1638-49.
[http://dx.doi.org/10.1016/j.addr.2008.08.002] [PMID: 18840488] -8Hennink WE, Van Nostrum C. Novel crosslinking methods to design hydrogels. Adv Drug Deliv Rev 2012; 64: 223-36.
[http://dx.doi.org/10.1016/j.addr.2012.09.009] [PMID: 11755704] ].
In recent years, the quest for materials-based hydrogels with potent functionalities including antimicrobial potentialities has revitalized the field of biomaterials. Fig. (1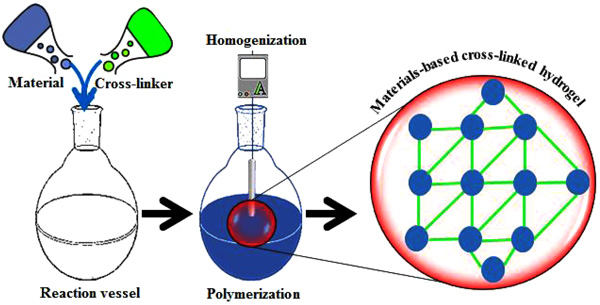 ) illustrates a simplified synthesis of the materials-based hydrogel in which a material of interest is polymerized in the presence of a functional cross-linking agent. Whereas, a bio-responsive behavior of control drug (antimicrobial/antiviral) release subject to pH and temperature change is shown in Fig. (2
) illustrates a simplified synthesis of the materials-based hydrogel in which a material of interest is polymerized in the presence of a functional cross-linking agent. Whereas, a bio-responsive behavior of control drug (antimicrobial/antiviral) release subject to pH and temperature change is shown in Fig. (2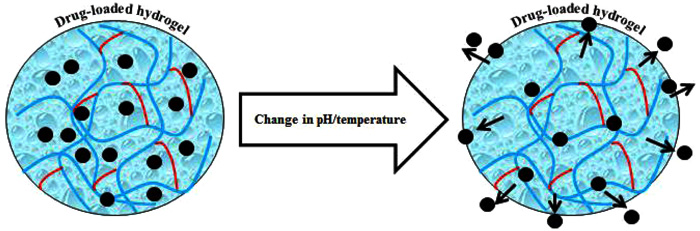 ) [3Villalba-Rodriguez AM, Dhama K, Iqbal HMN. Biomaterials-based hydrogels and their drug delivery potentialities. Int J Pharmacol 2017; 13(7): 864-73.
) [3Villalba-Rodriguez AM, Dhama K, Iqbal HMN. Biomaterials-based hydrogels and their drug delivery potentialities. Int J Pharmacol 2017; 13(7): 864-73.
[http://dx.doi.org/10.3923/ijp.2017.864.873] ]. With ever-increasing scientific awareness, novel approaches and technologies in the hydrogel design have been introduced that offer with significantly improved mechanical properties, super porous, and comb-type grafted hydrogels [9Yoshida R, Uchida K, Kaneko Y, Sakai K. Comb-type grafted hydrogels with rapid de-swelling response to temperature changes. Nature 1995; 374(6519): 240.
[http://dx.doi.org/10.1038/374240a0] -14Haraguchi K, Li HJ, Okumura N. Hydrogels with hydrophobic surfaces: Abnormally high contact angles for water on PNIPA nanocomposite hydrogels. Macromolecules 2007; 40(7): 2299-302.
[http://dx.doi.org/10.1021/ma062965u] ]. Other examples of hydrogel biomaterials with a smart future include, self-assembled hydrogels with tunable protein domains, stimuli (external/internal) responsive hydrogels, and genetically engineered triblock copolymers based hydrogels [15Petka WA, Harden JL, McGrath KP, Wirtz D, Tirrell DA. Reversible hydrogels from self-assembling artificial proteins. Science 1998; 281(5375): 389-92.
[http://dx.doi.org/10.1126/science.281.5375.389] [PMID: 9665877] -18Yang J, Xu C, Wang C, Kopeček J. Refolding hydrogels self-assembled from N-(2-hydroxypropyl)methacrylamide graft copolymers by antiparallel coiled-coil formation. Biomacromolecules 2006; 7(4): 1187-95.
[http://dx.doi.org/10.1021/bm051002k] [PMID: 16602737] ].
 |
Fig. (1) Schematic representation of materials-based hydrogel preparation in the presence of the functional cross-linking agent. |
 |
Fig. (2) The bio-responsive behavior of control drug release subject to pH and temperature change. The drug molecules could be antimicrobial or antiviral depending on the requirement (Reproduced from Ref [3Villalba-Rodriguez AM, Dhama K, Iqbal HMN. Biomaterials-based hydrogels and their drug delivery potentialities. Int J Pharmacol 2017; 13(7): 864-73. [http://dx.doi.org/10.3923/ijp.2017.864.873] ], an open-access article distributed under the terms of the creative commons attribution License). |
The present review work focuses on the development of novel types of materials-based hydrogels with antimicrobial and antiviral potentialities for various biomedical sectors of the modern world. Herein, this review is not presenting the whole literature about biomaterials as it is a big field with huge information. Thus, the focus is only given to the hydrogels perspective. The AMR/MDR resistance issues along with viral infections have been highlighted along with the possible action mechanisms of the hydrogels are briefly discussed with a closeup look at the future recommendations. Towards the end, outstanding issues posing questions mark and needs to be addressed are presented that can pave the way for future studies.
2. BIOMATERIALS - A BIOMIMETIC APPROACH
The development of new types of novel, effective and highly reliable materials-based hydrogels for multipurpose applications is essential and a core demand to tackle many human health related diseases. Owing to the unique chemical structure, bioactivity, non-toxicity, biocompatibility, biodegradability, recyclability, etc. bio-based materials possesses several complementary functionalities that position them well in the materials sector of the modern world. Many bio-based materials including chitin, chitosan, bacterial cellulose, alginate, and keratin, among others have been fully characterized and well organized/developed into value-added structures [19Iqbal HMN. Development of bio-composites with novel characteristics through enzymatic grafting 2015. Available from: http:// westminster research.wmin.ac.uk/ 16355/-33Rasheed T, Bilal M, Abu-Thabit NY, Iqbal HM. The smart chemistry of stimuli-responsive polymeric carriers for target drug delivery applications. In: Abdel Salam Hamdy Makhlouf ASH, Abu-Thabit NY, Eds. Stimuli Responsive Polymeric Nanocarriers for Drug Delivery Applications, Woodhead Publishing Series in Biomaterials 2018; 1: 61-99.
[http://dx.doi.org/10.1016/B978-0-08-101997-9.00003-5] ]. Thus, provide a proper route to emulate bio-systems - a biomimetic approach to eliminate the concerns like unfavorable immune responses, disease transmission, effective wound healing, control delivery and regeneration potentialities [34Iqbal HMN, Dhama K, Munjal A, et al. Recent trends in nanotechnology-based drugs and formulations for targeted therapeutic delivery. Recent Pat Inflamm Allergy Drug Discov 2016; 10(2): 86-93., 35Iqbal HMN, Dhama K, Munjal A, et al. Tissue engineering and regenerative medicine potentialities of materials-based novel constructs - A review. Curr Regen Med 2016; 6: 29-40.
[http://dx.doi.org/10.2174/2468424407666170210144332] ].
3. AMR/MDR RESISTANCE ISSUES
With ever increasing scientific knowledge and social awareness, now the people are more concern about the AMR/MDR issues. The increasing challenge to health care attributable to the AMR/MDR, therefore, AMR has become a worldwide concern, in recent years [36Jindal AK, Pandya K, Khan ID. Antimicrobial resistance: A public health challenge. Med J Armed Forces India 2015; 71(2): 178-81.
[http://dx.doi.org/10.1016/j.mjafi.2014.04.011] [PMID: 25859082] -41Rasheed T, Bilal M, Iqbal HMN, Li C. Green biosynthesis of silver nanoparticles using leaves extract of Artemisia vulgaris and their potential biomedical applications. Colloids Surf B Biointerfaces 2017; 158: 408-15.
[http://dx.doi.org/10.1016/j.colsurfb.2017.07.020] [PMID: 28719862] ]. Broadly speaking, AMR is defined as a temporary and/or permanent capability of a microbial strain and its progeny to resist and/or stay viable and multiply against the medication previously used to treat them. Owing to this notable resistivity and non-susceptibility, microbes have been classified as resistant strains to the concentration of an antimicrobial agent used in practice [42Cloete TE. Resistance mechanisms of bacteria to antimicrobial compounds. Int Biodeterior Biodegradation 2003; 51(4): 277-82.
[http://dx.doi.org/10.1016/S0964-8305(03)00042-8] ]. The AMR/MDR is a growing problem at the global level. Developing a range of strategies to reduce reliance on antimicrobials will be a key challenge for the future [43 UK Five Year Antimicrobial Resistance Strategy 2013 to 2018 (PDF). Available from:
https:// www.gov.uk/ government/ uploads/ system/ uploads/ attachment_data/ file/244058/ 20130902_UK_5_year_AMR_strategy.pdf [Last accessed: October 04, 2016].]. Owing to the antibiotic-resistant, infections now account for 25,000 deaths in Europe alone (European Centre for Disease Prevention and Control), and about 23,000 deaths and over 2 million illnesses in the US (Centers for Disease Control and Prevention), annually. In September 2014, the US President “Obama” signed an Executive Order instructing key health agencies to take action to combat the rise of antibiotic-resistant bacteria [44 https://www.whitehouse.gov/the-press-office/2014/09/18/fact-sheet-obama-administration-takes-actions-combat-antibiotic-resistan [Last accessed: October 04, 2016].]. Owing to the emerging or re-emerging infectious diseases caused by various microorganisms, much attention is now being focused towards alternative approaches to control and/or limit such deadly infections. In this context, novel materials with antimicrobial activities are attracting the considerable attention of both academia and industry, especially in the biomedical, and other health-related sectors of the modern world [19Iqbal HMN. Development of bio-composites with novel characteristics through enzymatic grafting 2015. Available from: http:// westminster research.wmin.ac.uk/ 16355/, 23Iqbal HMN, Kyazze G, Locke IC, Tron T, Keshavarz T. In situ development of self-defensive antibacterial biomaterials: Phenol-g-keratin-EC based bio-composites with characteristics for biomedical applications. Green Chem 2015; 17(7): 3858-69.
[http://dx.doi.org/10.1039/C5GC00715A] -25Iqbal HMN, Kyazze G, Locke IC, Tron T, Keshavarz T. Development of bio-composites with novel characteristics: Evaluation of phenol-induced antibacterial, biocompatible and biodegradable behaviours. Carbohydr Polym 2015; 131: 197-207.
[http://dx.doi.org/10.1016/j.carbpol.2015.05.046] [PMID: 26256176] , 27Iqbal HMN, Kyazze G, Locke IC, Tron T, Keshavarz T. Poly(3-hydroxybutyrate)-ethyl cellulose based bio-composites with novel characteristics for infection free wound healing application. Int J Biol Macromol 2015; 81: 552-9.
[http://dx.doi.org/10.1016/j.ijbiomac.2015.08.040] [PMID: 26314909] ]. Because of the growing consciousness and demands of legislative authorities, the manufacture, to reduce bacterial population in healthcare facilities and possibly to cut pathogenic infections, development of novel anti-microbial active materials which are biocompatible and biodegradable are considered to be a potential solution to such a problematic issue.
Among the potential causes, below are some possible explanations for an increased AMR/MDR:
- The genetic transformation from strain to strain.
- Biofilm matrix forming potential of several strains.
- Efflux pumps and other outer membrane structural variations.
- Enzyme-mediated resistance against, in practice, antimicrobials.
- Enhanced level of metabolic activity within the biofilm structure.
- Lower/no perfusion of antimicrobial agents through the biofilm matrix.
- Adaptability and interaction between antimicrobial agents and biofilm matrix.
- Excessive/useless consumption of in practice antimicrobials in a random order.
- Genetic variation and adaptability against the excessive antimicrobials exposure.
4. CLASSIFICATION OF HYDROGELS
Based on the literature data, hydrogels have been classified in several ways depending on the preparatory techniques, materials sources, ionic charges, stimuli (external or internal) responsiveness, crosslinking nature and biodegradability characteristics, etc. Though each method has its specificity and uniqueness, however, among all of the ways mentioned above, one of the important classifications is based on their crosslinking nature (Fig. 3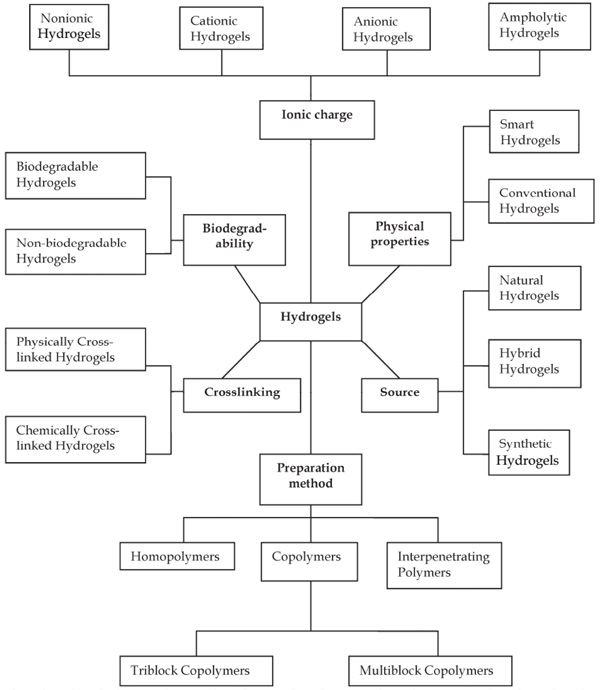 ) [44 https://www.whitehouse.gov/the-press-office/2014/09/18/fact-sheet-obama-administration-takes-actions-combat-antibiotic-resistan [Last accessed: October 04, 2016].-48Patel A, Mequanint K. Hydrogel biomaterials.Biomedical engineering-frontiers and challenges 2011; 275-96.
) [44 https://www.whitehouse.gov/the-press-office/2014/09/18/fact-sheet-obama-administration-takes-actions-combat-antibiotic-resistan [Last accessed: October 04, 2016].-48Patel A, Mequanint K. Hydrogel biomaterials.Biomedical engineering-frontiers and challenges 2011; 275-96.
[http://dx.doi.org/10.5772/24856] ]. The crosslinking nature either chemical or physical plays a critical role in the network stability of hydrogels in their swollen state [48Patel A, Mequanint K. Hydrogel biomaterials.Biomedical engineering-frontiers and challenges 2011; 275-96.
[http://dx.doi.org/10.5772/24856] ]. A detailed classification of hydrogels along with the preparation and potential applications of each sub-classified type are reviewed elsewhere [1Berger J, Reist M, Mayer JM, Felt O, Peppas NA, Gurny R. Structure and interactions in covalently and ionically crosslinked chitosan hydrogels for biomedical applications. Eur J Pharm Biopharm 2004; 57(1): 19-34.
[http://dx.doi.org/10.1016/S0939-6411(03)00161-9] [PMID: 14729078] , 48Patel A, Mequanint K. Hydrogel biomaterials.Biomedical engineering-frontiers and challenges 2011; 275-96.
[http://dx.doi.org/10.5772/24856] -55Bahram M, Mohseni N, Moghtader M. An introduction to hydrogels and some recent applications.Emerging Concepts in Analysis and Applications of Hydrogels 2016; 9-38.
[http://dx.doi.org/10.5772/64301] ], thus not the focus of this review.
 |
Fig. (3) Classification of Hydrogels (Reproduced from Ref [48Patel A, Mequanint K. Hydrogel biomaterials.Biomedical engineering-frontiers and challenges 2011; 275-96. [http://dx.doi.org/10.5772/24856] ], an open access article distributed under the terms of the creative commons attribution License). |
5. TECHNOLOGICAL FEATURES OF HYDROGELS
Owing to the diversity, hydrogels offer numerous technical and functional features. For example:
- Overall cost-effective ratio
- Minimal residual monomer
- Maximal absorption capacity
- Colorlessness, odourlessness
- Swelling and de-swelling property
- Tunable particle size and porosity
- Stability, compatibility, and biodegradability
The above-listed features are just considerable examples and are tunable depending on the materials used and requisite application.
6. HYDROGELS WITH ANTIMICROBIAL POTENTIALITIES
Hydrogels with antimicrobial potentialities are envisioned to be a fundamental weapon to combat AMR/MDR infections. Recently, the emergence of AMR/MDR microbial strains has created an innumerable challenge within the healthcare arena. Much sadly, current problems associated with the AMR/MDR microbial strains outspread far beyond gram-positive bacteria such as MRSA [56Li Y, Fukushima K, Coady DJ, et al. Broad-spectrum antimicrobial and biofilm-disrupting hydrogels: Stereocomplex-driven supramolecular assemblies. Angew Chem Int Ed Engl 2013; 52(2): 674-8.
[http://dx.doi.org/10.1002/anie.201206053] [PMID: 23161823] ]. In this context, there is a demanding need to engineer antimicrobial active hydrogels using above mentioned materials. As discussed above, the antimicrobial is a requisite property, and the material e.g. hydrogel should possess the following characteristics [57Kenawy R, Worley SD, Broughton R. The chemistry and applications of antimicrobial polymers: A state-of-the-art review. Biomacromolecules 2007; 8(5): 1359-84.
[http://dx.doi.org/10.1021/bm061150q] [PMID: 17425365] ].
- Eco-friendlier processing conditions
- Highly stable with a long-term shelf life
- Does not generate toxic products/byproduct, while in use
- High level of biocidal against a wider spectrum of pathogenic microbes
Among many potent materials, chitosan as a natural polyaminosaccharide possesses many of the above-attributed features. Thomas et al. [58Thomas V, Yallapu MM, Sreedhar B, Bajpai SK. A versatile strategy to fabricate hydrogel-silver nanocomposites and investigation of their antimicrobial activity. J Colloid Interface Sci 2007; 315(1): 389-95.
[http://dx.doi.org/10.1016/j.jcis.2007.06.068] [PMID: 17707388] ] reported a facile in-situ procedure to fabricate hydrogel–silver nanocomposites and investigation of their antimicrobial activity. The process involves the formation of silver nanoparticles within swollen poly (acrylamide-co-acrylic acid) hydrogels. In the same study, the authors have demonstrated excellent antibacterial effects of the developed hydrogel–silver nanocomposites against Escherichia coli. Antibacterial activity of a chitosan–γ-poly (glutamic acid) polyelectrolyte hydrogel has been reported by Tsao and co-workers [59Tsao CT, Chang CH, Lin YY, et al. Antibacterial activity and biocompatibility of a chitosan-γ-poly(glutamic acid) polyelectrolyte complex hydrogel. Carbohydr Res 2010; 345(12): 1774-80.
[http://dx.doi.org/10.1016/j.carres.2010.06.002] [PMID: 20598293] ]. The developed hydrogel was comprised on chitosan as the cationic polyelectrolyte and γ-poly(glutamic acid) (γ-PGA) as the anionic polyelectrolyte. The chitosan–γ-PGA hydrogels exhibited antibacterial activity against Escherichia coli and Staphylococcus aureus. Later, the same group of authors have evaluated the chitosan/γ-poly (glutamic acid) polyelectrolyte complex hydrogels and proved useful for wound-healing capabilities [60Tsao CT, Chang CH, Lin YY, et al. Evaluation of chitosan/γ-poly (glutamic acid) polyelectrolyte complex for wound dressing materials. Carbohydr Polym 2011; 84(2): 812-9.
[http://dx.doi.org/10.1016/j.carbpol.2010.04.034] ]. Though chitosan is well known to exhibit considerable antibacterial activity against different microbial strains, however, there are some limitations regarding its molecular weight. Based on the literature data, the chitosan with low molecular weight has higher/stronger biocidal/antibacterial activity against Escherichia coli than that of the chitosan with higher molecular weight [61Liu N, Chen XG, Park HJ, et al. Effect of MW and concentration of chitosan on antibacterial activity of Escherichia coli. Carbohydr Polym 2006; 64(1): 60-5.
[http://dx.doi.org/10.1016/j.carbpol.2005.10.028] ]. Likewise, in an earlier study, No et al. [62No HK, Park NY, Lee SH, Meyers SP. Antibacterial activity of chitosans and chitosan oligomers with different molecular weights. Int J Food Microbiol 2002; 74(1-2): 65-72.
[http://dx.doi.org/10.1016/S0168-1605(01)00717-6] [PMID: 11929171] ] reported antibacterial activities of six chitosans and six chitosan oligomers with different molecular weights against four gram-negative strains i.e.Escherichia coli, Pseudomonas fluorescens, Salmonella typhimurium, and Vibrio parahaemolyticus and seven gram-positive strains i.e.Listeria monocytogenes, Bacillus megaterium, B. cereus, Staphylococcus aureus, Lactobacillus plantarum, L. Brevis, and L. bulgaricus. The development of a novel hydrogel based on dimethyl alkyl ammonium chitosan (with various degrees of quaternization)-graft-poly(ethylene glycol) methacrylate (qC-g-EM) and poly(ethylene glycol) diacrylate with potent antibacterial activity have been reported [63Li P, Poon YF, Li W, et al. A polycationic antimicrobial and biocompatible hydrogel with microbe membrane suctioning ability. Nat Mater 2011; 10(2): 149-56.
[http://dx.doi.org/10.1038/nmat2915] [PMID: 21151166] ]. The reported hydrogel has excellent antimicrobial efficacy against Pseudomonas aeruginosa, Escherichia coli, Staphylococcus aureus and Fusarium solani. In the same study, authors reported that increasing the alkyl chain length of the quaternizing agent from Trimethylammonium (TM) to dimethyldecylammonium (DMD) led to greater efficacy against Gram-positive strain i.e.S. aureus but not the Gram-negative strain i.e.E. coli and P. aeruginosa.
7. ANTIMICROBIAL MECHANISM OF HYDROGELS
The antimicrobial mechanism of hydrogels from the materials with intrinsic antimicrobial activity is not fully understood. Various authors have proposed and justified antimicrobial mechanism of materials or materials based hydrogels and constructs [19Iqbal HMN. Development of bio-composites with novel characteristics through enzymatic grafting 2015. Available from: http:// westminster research.wmin.ac.uk/ 16355/, 23Iqbal HMN, Kyazze G, Locke IC, Tron T, Keshavarz T. In situ development of self-defensive antibacterial biomaterials: Phenol-g-keratin-EC based bio-composites with characteristics for biomedical applications. Green Chem 2015; 17(7): 3858-69.
[http://dx.doi.org/10.1039/C5GC00715A] -25Iqbal HMN, Kyazze G, Locke IC, Tron T, Keshavarz T. Development of bio-composites with novel characteristics: Evaluation of phenol-induced antibacterial, biocompatible and biodegradable behaviours. Carbohydr Polym 2015; 131: 197-207.
[http://dx.doi.org/10.1016/j.carbpol.2015.05.046] [PMID: 26256176] , 27Iqbal HMN, Kyazze G, Locke IC, Tron T, Keshavarz T. Poly(3-hydroxybutyrate)-ethyl cellulose based bio-composites with novel characteristics for infection free wound healing application. Int J Biol Macromol 2015; 81: 552-9.
[http://dx.doi.org/10.1016/j.ijbiomac.2015.08.040] [PMID: 26314909] , 38Bilal M, Rasheed T, Iqbal HMN, Hu H, Wang W, Zhang X. Macromolecular agents with antimicrobial potentialities: A drive to combat antimicrobial resistance. Int J Biol Macromol 2017; 103: 554-74.
[http://dx.doi.org/10.1016/j.ijbiomac.2017.05.071] [PMID: 28528940] , 63Li P, Poon YF, Li W, et al. A polycationic antimicrobial and biocompatible hydrogel with microbe membrane suctioning ability. Nat Mater 2011; 10(2): 149-56.
[http://dx.doi.org/10.1038/nmat2915] [PMID: 21151166] -66Li Y, Fukushima K, Coady DJ, et al. Broad-spectrum antimicrobial and biofilm-disrupting hydrogels: Stereocomplex-driven supramolecular assemblies. Angew Chem Int Ed Engl 2013; 52(2): 674-8.
[http://dx.doi.org/10.1002/anie.201206053] [PMID: 23161823] ]. Most of the reported antimicrobial mechanisms are based on the bacterial cell membrane lysis subject to the available reactive functional groups and routes. The anionic bacterial cells are attracted to the cationic materials or materials based hydrogels through electrostatic interaction, leading to subsequent cell wall disruption, membrane lysis, cell leakage that ultimately leads to cell death. In summary, the following points play major/critical role in the antimicrobial activity of materials or materials based hydrogels.
- Charge (cationic) surface ratio
- Amphiphilicity of the materials
- The available alkyl chain length
- Availability of reactive functional groups
- Overall pore size of the materials final product
- Permeability features of the target (microbial) membrane
8. HYDROGELS WITH ANTIVIRAL POTENTIALITIES
In practice antiviral therapeutics e.g. oseltamivir and zanamivir, etc. are facing increasing problems with resistance development. Oseltamivir is a selective antiviral prodrug which is used to tackle influenza virus. Whereas, zanamivir is an inhibitor of neuraminidase used in the treatment of common flu and in the prophylaxis of virus A and B. Engineering efficient antiviral drugs with potent activities against a wider spectrum of viral pathogens is difficult because viruses use the host's cells to replicate. Therefore, researchers, around the globe, are working to extend the range of antivirals to other families of pathogens. Owing to the ever-increasing drug resistance, there is an urgent need to develop novel formulations in a range of contexts to tackle various viral infections. Furthermore, the constantly changing genetic makeup of viruses may alter or induce the viral resistance against several in-practice treatment strategies [67Nijhuis M, Van Maarseveen NM, Boucher CAB. Antiviral resistance and impact on viral replication capacity: Evolution of viruses under antiviral pressure occurs in three phases.Antiviral Strategies 2009; 299-320.
[http://dx.doi.org/10.1007/978-3-540-79086-0_11] ]. Spontaneous or intermittent mechanisms are mainly responsible for viral resistant throughout the antiviral treatment. In an earlier study, Herlocher et al. [68Herlocher ML, Truscon R, Elias S, et al. Influenza viruses resistant to the antiviral drug oseltamivir: Transmission studies in ferrets. J Infect Dis 2004; 190(9): 1627-30.
[http://dx.doi.org/10.1086/424572] [PMID: 15478068] ] isolated three type A influenza viruses, each of which has a distinct neuraminidase-gene mutation and is resistant to the neuraminidase inhibitor oseltamivir. Likewise, immunocompromised patients, who received oseltamivir for “post-exposure prophylaxis” are also at higher risk of resistance [69WHO. 2009.Antiviral use and the risk of drug resistance Available from:
http://www.who.int/csr/disease/swineflu/notes/h1n1 _antiviral_use_20090925/en].
In this context, biomaterials-based hydrogels with antimicrobial “non-drugs” e.g. nanoparticles find widespread biomedical applications [70Malmsten M. Antimicrobial and antiviral hydrogels. Soft Matter 2011; 7(19): 8725-36.
[http://dx.doi.org/10.1039/c1sm05809f] ]. Such biomaterials-based hydrogels systems with incorporated compounds offer additional advantages e.g. low or no toxicity, responsive behavior, favorable tissue integration, targeted and control drug release potentialities, and controlled degradation rate. With these added values, considerable research efforts have been made to incorporate antiviral drugs into hydrogels. For example, Thorgeirsdottir et al. [71Thorgeirsdóttir TO, Thormar H, Kristmundsdóttir T. Effects of polysorbates on antiviral and antibacterial activity of monoglyceride in pharmaceutical formulations. Pharmazie 2003; 58(4): 286-7.
[PMID: 12749415] ] reported that for monocaprin solutions containing 5% propylene glycol, the antiviral activity is greatly reduced by 5% polysorbate 20. In the same study, authors have also revealed that solutions containing 7.5% propylene glycol and polysorbate 20 (0.75 to 1.5% concentration) were found to have antiviral activities comparable to that of pure monocaprin [71Thorgeirsdóttir TO, Thormar H, Kristmundsdóttir T. Effects of polysorbates on antiviral and antibacterial activity of monoglyceride in pharmaceutical formulations. Pharmazie 2003; 58(4): 286-7.
[PMID: 12749415] ]. Considering antiviral agents, monocaprin is one among the highly efficient monoglycerides. It has also been shown to be effective against the enveloped viruses e.g. vesicular stomatitis virus, Herpes Simplex Virus (HSV), visna virus and human immunodeficiency virus in-vitro [72Kristmundsdóttir T, Arnadóttir SG, Bergsson G, Thormar H. Development and evaluation of microbicidal hydrogels containing monoglyceride as the active ingredient. J Pharm Sci 1999; 88(10): 1011-5.
[http://dx.doi.org/10.1021/js9900396] [PMID: 10514348] , 73Thormar H, Bergsson G, Gunnarsson E, et al. Hydrogels containing monocaprin have potent microbicidal activities against sexually transmitted viruses and bacteria in vitro. Sex Transm Infect 1999; 75(3): 181-5.
[http://dx.doi.org/10.1136/sti.75.3.181] [PMID: 10448397] ]. Similarly, hydrophilic gels containing monocaprin in a concentration of 20 mM have shown more than 100,000-fold inactivation of HSV-2 and HSV-1 [72Kristmundsdóttir T, Arnadóttir SG, Bergsson G, Thormar H. Development and evaluation of microbicidal hydrogels containing monoglyceride as the active ingredient. J Pharm Sci 1999; 88(10): 1011-5.
[http://dx.doi.org/10.1021/js9900396] [PMID: 10514348] , 73Thormar H, Bergsson G, Gunnarsson E, et al. Hydrogels containing monocaprin have potent microbicidal activities against sexually transmitted viruses and bacteria in vitro. Sex Transm Infect 1999; 75(3): 181-5.
[http://dx.doi.org/10.1136/sti.75.3.181] [PMID: 10448397] ]. In another study, Rokhade et al. [74Rokhade AP, Patil SA, Aminabhavi TM. Synthesis and characterization of semi-interpenetrating polymer network microspheres of acrylamide grafted dextran and chitosan of acyclovir. Carbohydr Polym 2007; 67(4): 605-13.
[http://dx.doi.org/10.1016/j.carbpol.2006.07.001] ] developed semi-interpenetrating polymer network microspheres of acrylamide-grafted dextran and chitosan-based hydrogels for controlled release of antiviral drug acyclovir. Chiappetta et al. [75Chiappetta DA, Hocht C, Taira C, Sosnik A. Efavirenz-loaded polymeric micelles for pediatric anti-HIV pharmacotherapy with significantly higher oral bioavailability [corrected]. Nanomedicine (Lond) 2010; 5(1): 11-23.
[http://dx.doi.org/10.2217/nnm.09.90] [PMID: 20025460] , 76Chiappetta DA, Hocht C, Sosnik A. A highly concentrated and taste-improved aqueous formulation of efavirenz for a more appropriate pediatric management of the anti-HIV therapy. Curr HIV Res 2010; 8(3): 223-31.
[http://dx.doi.org/10.2174/157016210791111142] [PMID: 20158456] ] incorporated antiviral drug efavirenz into PEO/PPO block copolymer hydrogels for pediatric anti-HIV pharmacotherapy with significantly higher oral bioavailability.
9. CONCLUDING REMARKS AND FUTURE CONSIDERATIONS
In conclusion, this review highlights the potent features of materials based hydrogels possessing antimicrobial and antiviral potentialities. Herein, the work was aimed to critically overview the literature to establish an infective capacity of the naturally occurring materials or materials based hydrogels using a range of microbiological techniques against viral infections and various gram-positive and gram-negative pathogenic microbial strains including antibiotic-resistant forms. Through cautious strategy, the tunable materials or materials based hydrogels with multi-functionalities can be engineered to achieve optimal infective capability and therefore enhanced AMR/MDR control. Moreover, a novel type of potent materials could be designed for the management and skin regeneration/repair from injury, particularly burns and ulcers, where the risk of bacterial infection is high. Material structure and performance integrity need to be accessed using a range of analytical and imaging techniques.
Aiming to treat or tackle microbial and/or viral infections, a precise and control delivery of engineered constructs with diverse bioactivities is critical. The biocidal agents must be released from the gels to the target site to kill/eradicate the infectious agents in a safer and sustained manner. Such careful practices are essential to treat infections effectively to prevent biofilm formation that can play a crucial role in the development of resistant strains. Against, AMR/MDR strains, a strong synergistic effect can be achieved by engineering hydrogels using materials with intrinsic antimicrobial potential along with the impregnation of conventional antimicrobial agents.
10. OUTSTANDING QUESTIONS AND RESEARCH GAPS
Despite the huge research and plethora of reported literature on materials-based constructs including hydrogels, there are still outstanding issues posing questions mark and needs to address.
- Is it a cost-effective process to develop gels using multi-materials co-supported with other conventional antimicrobial agents?
- What could justify the development of gels which are active only against a certain number of strains instead of the wider-spectrum ones?
- What is the maximum alkyl chain length for the generation of a bactericidal efficacy at an optimal level?
- How can a robust methodology be designed to standardize the information and satisfy regulatory concerns?
- What are the ultimate and long-end consequences of the increased use of conventional antimicrobial agents?
- What are the health and environmental impacts of the heavy consumption of natural materials?
CONSENT FOR PUBLICATION
Not applicable.
CONFLICT OF INTEREST
The author declares no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
The literature facilities provided by Tecnologico de Monterrey, Mexico are thankfully acknowledged.
REFERENCES
| [1] | Berger J, Reist M, Mayer JM, Felt O, Peppas NA, Gurny R. Structure and interactions in covalently and ionically crosslinked chitosan hydrogels for biomedical applications. Eur J Pharm Biopharm 2004; 57(1): 19-34. [http://dx.doi.org/10.1016/S0939-6411(03)00161-9] [PMID: 14729078] |
| [2] | Kalshetti PP, Rajendra VB, Dixit DN, Parekh PP. Hydrogels as a drug delivery system and applications: A review. Int J Pharm Pharm Sci 2012; 4(1): 1-7. |
| [3] | Villalba-Rodriguez AM, Dhama K, Iqbal HMN. Biomaterials-based hydrogels and their drug delivery potentialities. Int J Pharmacol 2017; 13(7): 864-73. [http://dx.doi.org/10.3923/ijp.2017.864.873] |
| [4] | Iqbal HMN. Biomaterials: A trendy source to engineer functional entities-An overview. arXiv preprint 2018; arXiv:1803.05972. Available from: https://arxiv.org/abs/1803.05972 |
| [5] | Pal K, Banthia AK, Majumdar DK. Polymeric hydrogels: Characterization and biomedical applications. Des Monomers Polym 2009; 12(3): 197-220. [http://dx.doi.org/10.1163/156855509X436030] |
| [6] | Hamidi M, Azadi A, Rafiei P. Hydrogel nanoparticles in drug delivery. Adv Drug Deliv Rev 2008; 60(15): 1638-49. [http://dx.doi.org/10.1016/j.addr.2008.08.002] [PMID: 18840488] |
| [7] | Bhattarai N, Gunn J, Zhang M. Chitosan-based hydrogels for controlled, localized drug delivery. Adv Drug Deliv Rev 2010; 62(1): 83-99. [http://dx.doi.org/10.1016/j.addr.2009.07.019] [PMID: 19799949] |
| [8] | Hennink WE, Van Nostrum C. Novel crosslinking methods to design hydrogels. Adv Drug Deliv Rev 2012; 64: 223-36. [http://dx.doi.org/10.1016/j.addr.2012.09.009] [PMID: 11755704] |
| [9] | Yoshida R, Uchida K, Kaneko Y, Sakai K. Comb-type grafted hydrogels with rapid de-swelling response to temperature changes. Nature 1995; 374(6519): 240. [http://dx.doi.org/10.1038/374240a0] |
| [10] | Chen J, Park H, Park K. Synthesis of superporous hydrogels: Hydrogels with fast swelling and superabsorbent properties. J Biomed Mater Res 1999; 44(1): 53-62. [http://dx.doi.org/10.1002/(SICI)1097-4636(199901)44:1<53::AID-JBM6>3.0.CO;2-W] [PMID: 10397904] |
| [11] | Okumura Y, Ito K. The polyrotaxane gel: A topological gel by figure-of-eight cross-links. Adv Mater 2001; 13(7): 485-7. [http://dx.doi.org/10.1002/1521-4095(200104)13:7<485::AID-ADMA485>3.0.CO;2-T] |
| [12] | Haraguchi K, Takehisa T. Nanocomposite hydrogels: A unique organic-inorganic network structure with extraordinary mechanical, optical, and swelling/de-swelling properties. Adv Mater 2002; 14(16): 1120. [http://dx.doi.org/10.1002/1521-4095(20020816)14:16<1120::AID-ADMA1120>3.0.CO;2-9] |
| [13] | Gong JP, Katsuyama Y, Kurokawa T, Osada Y. Double network hydrogels with extremely high mechanical strength. Adv Mater 2003; 15(14): 1155-8. [http://dx.doi.org/10.1002/adma.200304907] |
| [14] | Haraguchi K, Li HJ, Okumura N. Hydrogels with hydrophobic surfaces: Abnormally high contact angles for water on PNIPA nanocomposite hydrogels. Macromolecules 2007; 40(7): 2299-302. [http://dx.doi.org/10.1021/ma062965u] |
| [15] | Petka WA, Harden JL, McGrath KP, Wirtz D, Tirrell DA. Reversible hydrogels from self-assembling artificial proteins. Science 1998; 281(5375): 389-92. [http://dx.doi.org/10.1126/science.281.5375.389] [PMID: 9665877] |
| [16] | Wang C, Stewart RJ, Kopecek J. Hybrid hydrogels assembled from synthetic polymers and coiled-coil protein domains. Nature 1999; 397(6718): 417-20. [http://dx.doi.org/10.1038/17092] [PMID: 9989405] |
| [17] | Xu C, Breedveld V, Kopeček J. Reversible hydrogels from self-assembling genetically engineered protein block copolymers. Biomacromolecules 2005; 6(3): 1739-49. [http://dx.doi.org/10.1021/bm050017f] [PMID: 15877401] |
| [18] | Yang J, Xu C, Wang C, Kopeček J. Refolding hydrogels self-assembled from N-(2-hydroxypropyl)methacrylamide graft copolymers by antiparallel coiled-coil formation. Biomacromolecules 2006; 7(4): 1187-95. [http://dx.doi.org/10.1021/bm051002k] [PMID: 16602737] |
| [19] | Iqbal HMN. Development of bio-composites with novel characteristics through enzymatic grafting 2015. Available from: http:// westminster research.wmin.ac.uk/ 16355/ |
| [20] | Iqbal HMN, Kyazze G, Tron T, Keshavarz T. “One-pot” synthesis and characterisation of novel P (3HB)-ethyl cellulose based graft composites through lipase catalysed esterification. Polym Chem 2014; 5(24): 7004-12. [http://dx.doi.org/10.1039/C4PY00857J] |
| [21] | Iqbal HMN, Kyazze G, Tron T, Keshavarz T. A preliminary study on the development and characterisation of enzymatically grafted P (3HB)-ethyl cellulose based novel composites. Cellulose 2014; 21(5): 3613-21. [http://dx.doi.org/10.1007/s10570-014-0337-9] |
| [22] | Iqbal HMN, Kyazze G, Tron T, Keshavarz T. Laccase-assisted grafting of poly(3-hydroxybutyrate) onto the bacterial cellulose as backbone polymer: Development and characterisation. Carbohydr Polym 2014; 113: 131-7. [http://dx.doi.org/10.1016/j.carbpol.2014.07.003] [PMID: 25256467] |
| [23] | Iqbal HMN, Kyazze G, Locke IC, Tron T, Keshavarz T. In situ development of self-defensive antibacterial biomaterials: Phenol-g-keratin-EC based bio-composites with characteristics for biomedical applications. Green Chem 2015; 17(7): 3858-69. [http://dx.doi.org/10.1039/C5GC00715A] |
| [24] | Iqbal HMN, Kyazze G, Locke IC, Tron T, Keshavarz T. Development of novel antibacterial active, HaCaT biocompatible and biodegradable CA-gP (3HB)-EC biocomposites with caffeic acid as a functional entity. Express Polym Lett 2015; 9: 764-72. [http://dx.doi.org/10.3144/expresspolymlett.2015.72] |
| [25] | Iqbal HMN, Kyazze G, Locke IC, Tron T, Keshavarz T. Development of bio-composites with novel characteristics: Evaluation of phenol-induced antibacterial, biocompatible and biodegradable behaviours. Carbohydr Polym 2015; 131: 197-207. [http://dx.doi.org/10.1016/j.carbpol.2015.05.046] [PMID: 26256176] |
| [26] | Iqbal HMN, Kyazze G, Tron T, Keshavarz T. Laccase assisted approach to graft multifunctional materials of interest: Keratin EC based novel composites and their characterisation. Macromol Mater Eng 2015; 300(7): 712-20. [http://dx.doi.org/10.1002/mame.201500003] |
| [27] | Iqbal HMN, Kyazze G, Locke IC, Tron T, Keshavarz T. Poly(3-hydroxybutyrate)-ethyl cellulose based bio-composites with novel characteristics for infection free wound healing application. Int J Biol Macromol 2015; 81: 552-9. [http://dx.doi.org/10.1016/j.ijbiomac.2015.08.040] [PMID: 26314909] |
| [28] | Bedian L, Villalba-Rodríguez AM, Hernández-Vargas G, Parra-Saldivar R, Iqbal HMN. Bio-based materials with novel characteristics for tissue engineering applications - A review. Int J Biol Macromol 2017; 98: 837-46. [http://dx.doi.org/10.1016/j.ijbiomac.2017.02.048] [PMID: 28223133] |
| [29] | Ruiz-Ruiz F, Mancera-Andrade EI, Parra-Saldivar R, Keshavarz T, Iqbal HMN. Drug delivery and cosmeceutical applications of poly- lactic acid based novel constructs - A review. Curr Drug Metab 2017; 18(10): 914-25. [PMID: 28933269] |
| [30] | Villalba-Rodriguez AM, Parra-Saldivar R, Ahmed I, et al. Bio-inspired biomaterials and their drug delivery perspectives - A review. Curr Drug Metab 2017; 18(10): 893-904. [PMID: 28952433] |
| [31] | Iqbal HMN, Kyazze G, Tron T, Keshavarz T. Laccase from Aspergillus niger: A novel tool to graft multifunctional materials of interests and their characterization. Saudi J Biol Sci 2018; 25(3): 545-50. [http://dx.doi.org/10.1016/j.sjbs.2016.01.027] [PMID: 29686517] |
| [32] | Iqbal HMN, Keshavarz T. Bioinspired polymeric carriers for drug delivery applications. In: Abdel Salam Hamdy Makhlouf ASH, Abu-Thabit NY, Eds. Stimuli responsive polymeric nanocarriers for drug delivery applications, Woodhead publishing series in biomaterials 2018; 1: 377-404. [http://dx.doi.org/10.1016/B978-0-08-101997-9.00018-7] |
| [33] | Rasheed T, Bilal M, Abu-Thabit NY, Iqbal HM. The smart chemistry of stimuli-responsive polymeric carriers for target drug delivery applications. In: Abdel Salam Hamdy Makhlouf ASH, Abu-Thabit NY, Eds. Stimuli Responsive Polymeric Nanocarriers for Drug Delivery Applications, Woodhead Publishing Series in Biomaterials 2018; 1: 61-99. [http://dx.doi.org/10.1016/B978-0-08-101997-9.00003-5] |
| [34] | Iqbal HMN, Dhama K, Munjal A, et al. Recent trends in nanotechnology-based drugs and formulations for targeted therapeutic delivery. Recent Pat Inflamm Allergy Drug Discov 2016; 10(2): 86-93. |
| [35] | Iqbal HMN, Dhama K, Munjal A, et al. Tissue engineering and regenerative medicine potentialities of materials-based novel constructs - A review. Curr Regen Med 2016; 6: 29-40. [http://dx.doi.org/10.2174/2468424407666170210144332] |
| [36] | Jindal AK, Pandya K, Khan ID. Antimicrobial resistance: A public health challenge. Med J Armed Forces India 2015; 71(2): 178-81. [http://dx.doi.org/10.1016/j.mjafi.2014.04.011] [PMID: 25859082] |
| [37] | Holmes AH, Moore LS, Sundsfjord A, et al. Understanding the mechanisms and drivers of antimicrobial resistance. Lancet 2016; 387(10014): 176-87. [http://dx.doi.org/10.1016/S0140-6736(15)00473-0] [PMID: 26603922] |
| [38] | Bilal M, Rasheed T, Iqbal HMN, Hu H, Wang W, Zhang X. Macromolecular agents with antimicrobial potentialities: A drive to combat antimicrobial resistance. Int J Biol Macromol 2017; 103: 554-74. [http://dx.doi.org/10.1016/j.ijbiomac.2017.05.071] [PMID: 28528940] |
| [39] | Bilal M, Rasheed T, Iqbal HMN, Li C, Hu H, Zhang X. Development of silver nanoparticles loaded chitosan-alginate constructs with biomedical potentialities. Int J Biol Macromol 2017; 105(Pt 1): 393-400. [http://dx.doi.org/10.1016/j.ijbiomac.2017.07.047] [PMID: 28705499] |
| [40] | Bilal M, Rasheed T, Iqbal HMN, Hu H, Zhang X. Silver nanoparticles: Biosynthesis and antimicrobial potentialities. Int J Pharmacol 2017; 13(7): 832-45. [http://dx.doi.org/10.3923/ijp.2017.832.845] |
| [41] | Rasheed T, Bilal M, Iqbal HMN, Li C. Green biosynthesis of silver nanoparticles using leaves extract of Artemisia vulgaris and their potential biomedical applications. Colloids Surf B Biointerfaces 2017; 158: 408-15. [http://dx.doi.org/10.1016/j.colsurfb.2017.07.020] [PMID: 28719862] |
| [42] | Cloete TE. Resistance mechanisms of bacteria to antimicrobial compounds. Int Biodeterior Biodegradation 2003; 51(4): 277-82. [http://dx.doi.org/10.1016/S0964-8305(03)00042-8] |
| [43] | UK Five Year Antimicrobial Resistance Strategy 2013 to 2018 (PDF). Available from: https:// www.gov.uk/ government/ uploads/ system/ uploads/ attachment_data/ file/244058/ 20130902_UK_5_year_AMR_strategy.pdf [Last accessed: October 04, 2016]. |
| [44] | https://www.whitehouse.gov/the-press-office/2014/09/18/fact-sheet-obama-administration-takes-actions-combat-antibiotic-resistan [Last accessed: October 04, 2016]. |
| [45] | Dumitriu S, Ed. Polymeric Biomatrials 2002. |
| [46] | Hin T. Engineering Materials for Biomedical Applications 2004. [http://dx.doi.org/10.1142/5673] |
| [47] | Ratner BD, Hoffman AS, Schoen FJ, Lemons JE, Eds. Biomaterials science: An introduction to materials in medicine 2004. |
| [48] | Patel A, Mequanint K. Hydrogel biomaterials.Biomedical engineering-frontiers and challenges 2011; 275-96. [http://dx.doi.org/10.5772/24856] |
| [49] | Hoare TR, Kohane DS. Hydrogels in drug delivery: Progress and challenges. Polymer (Guildf) 2008; 49(8): 1993-2007. [http://dx.doi.org/10.1016/j.polymer.2008.01.027] |
| [50] | Bhattarai N, Gunn J, Zhang M. Chitosan-based hydrogels for controlled, localized drug delivery. Adv Drug Deliv Rev 2010; 62(1): 83-99. [http://dx.doi.org/10.1016/j.addr.2009.07.019] [PMID: 19799949] |
| [51] | Laftah WA, Hashim S, Ibrahim AN. Polymer hydrogels: A review. Polym Plast Technol Eng 2011; 50(14): 1475-86. [http://dx.doi.org/10.1080/03602559.2011.593082] |
| [52] | Ahmed EM. Hydrogel: Preparation, characterization, and applications: A review. J Adv Res 2015; 6(2): 105-21. [http://dx.doi.org/10.1016/j.jare.2013.07.006] [PMID: 25750745] |
| [53] | Caló E, Khutoryanskiy VV. Biomedical applications of hydrogels: A review of patents and commercial products. Eur Polym J 2015; 65: 252-67. [http://dx.doi.org/10.1016/j.eurpolymj.2014.11.024] |
| [54] | Ullah F, Othman MBH, Javed F, Ahmad Z, Md Akil H. Classification, processing and application of hydrogels: A review. Mater Sci Eng C 2015; 57: 414-33. [http://dx.doi.org/10.1016/j.msec.2015.07.053] [PMID: 26354282] |
| [55] | Bahram M, Mohseni N, Moghtader M. An introduction to hydrogels and some recent applications.Emerging Concepts in Analysis and Applications of Hydrogels 2016; 9-38. [http://dx.doi.org/10.5772/64301] |
| [56] | Li Y, Fukushima K, Coady DJ, et al. Broad-spectrum antimicrobial and biofilm-disrupting hydrogels: Stereocomplex-driven supramolecular assemblies. Angew Chem Int Ed Engl 2013; 52(2): 674-8. [http://dx.doi.org/10.1002/anie.201206053] [PMID: 23161823] |
| [57] | Kenawy R, Worley SD, Broughton R. The chemistry and applications of antimicrobial polymers: A state-of-the-art review. Biomacromolecules 2007; 8(5): 1359-84. [http://dx.doi.org/10.1021/bm061150q] [PMID: 17425365] |
| [58] | Thomas V, Yallapu MM, Sreedhar B, Bajpai SK. A versatile strategy to fabricate hydrogel-silver nanocomposites and investigation of their antimicrobial activity. J Colloid Interface Sci 2007; 315(1): 389-95. [http://dx.doi.org/10.1016/j.jcis.2007.06.068] [PMID: 17707388] |
| [59] | Tsao CT, Chang CH, Lin YY, et al. Antibacterial activity and biocompatibility of a chitosan-γ-poly(glutamic acid) polyelectrolyte complex hydrogel. Carbohydr Res 2010; 345(12): 1774-80. [http://dx.doi.org/10.1016/j.carres.2010.06.002] [PMID: 20598293] |
| [60] | Tsao CT, Chang CH, Lin YY, et al. Evaluation of chitosan/γ-poly (glutamic acid) polyelectrolyte complex for wound dressing materials. Carbohydr Polym 2011; 84(2): 812-9. [http://dx.doi.org/10.1016/j.carbpol.2010.04.034] |
| [61] | Liu N, Chen XG, Park HJ, et al. Effect of MW and concentration of chitosan on antibacterial activity of Escherichia coli. Carbohydr Polym 2006; 64(1): 60-5. [http://dx.doi.org/10.1016/j.carbpol.2005.10.028] |
| [62] | No HK, Park NY, Lee SH, Meyers SP. Antibacterial activity of chitosans and chitosan oligomers with different molecular weights. Int J Food Microbiol 2002; 74(1-2): 65-72. [http://dx.doi.org/10.1016/S0168-1605(01)00717-6] [PMID: 11929171] |
| [63] | Li P, Poon YF, Li W, et al. A polycationic antimicrobial and biocompatible hydrogel with microbe membrane suctioning ability. Nat Mater 2011; 10(2): 149-56. [http://dx.doi.org/10.1038/nmat2915] [PMID: 21151166] |
| [64] | Zhou C, Li P, Qi X, et al. A photopolymerized antimicrobial hydrogel coating derived from epsilon-poly-L-lysine. Biomaterials 2011; 32(11): 2704-12. [http://dx.doi.org/10.1016/j.biomaterials.2010.12.040] [PMID: 21257199] |
| [65] | Liu SQ, Yang C, Huang Y, et al. Antimicrobial and antifouling hydrogels formed in situ from polycarbonate and poly(ethylene glycol) via Michael addition. Adv Mater 2012; 24(48): 6484-9. [http://dx.doi.org/10.1002/adma.201202225] [PMID: 23018988] |
| [66] | Li Y, Fukushima K, Coady DJ, et al. Broad-spectrum antimicrobial and biofilm-disrupting hydrogels: Stereocomplex-driven supramolecular assemblies. Angew Chem Int Ed Engl 2013; 52(2): 674-8. [http://dx.doi.org/10.1002/anie.201206053] [PMID: 23161823] |
| [67] | Nijhuis M, Van Maarseveen NM, Boucher CAB. Antiviral resistance and impact on viral replication capacity: Evolution of viruses under antiviral pressure occurs in three phases.Antiviral Strategies 2009; 299-320. [http://dx.doi.org/10.1007/978-3-540-79086-0_11] |
| [68] | Herlocher ML, Truscon R, Elias S, et al. Influenza viruses resistant to the antiviral drug oseltamivir: Transmission studies in ferrets. J Infect Dis 2004; 190(9): 1627-30. [http://dx.doi.org/10.1086/424572] [PMID: 15478068] |
| [69] | WHO. 2009.Antiviral use and the risk of drug resistance Available from: http://www.who.int/csr/disease/swineflu/notes/h1n1 _antiviral_use_20090925/en |
| [70] | Malmsten M. Antimicrobial and antiviral hydrogels. Soft Matter 2011; 7(19): 8725-36. [http://dx.doi.org/10.1039/c1sm05809f] |
| [71] | Thorgeirsdóttir TO, Thormar H, Kristmundsdóttir T. Effects of polysorbates on antiviral and antibacterial activity of monoglyceride in pharmaceutical formulations. Pharmazie 2003; 58(4): 286-7. [PMID: 12749415] |
| [72] | Kristmundsdóttir T, Arnadóttir SG, Bergsson G, Thormar H. Development and evaluation of microbicidal hydrogels containing monoglyceride as the active ingredient. J Pharm Sci 1999; 88(10): 1011-5. [http://dx.doi.org/10.1021/js9900396] [PMID: 10514348] |
| [73] | Thormar H, Bergsson G, Gunnarsson E, et al. Hydrogels containing monocaprin have potent microbicidal activities against sexually transmitted viruses and bacteria in vitro. Sex Transm Infect 1999; 75(3): 181-5. [http://dx.doi.org/10.1136/sti.75.3.181] [PMID: 10448397] |
| [74] | Rokhade AP, Patil SA, Aminabhavi TM. Synthesis and characterization of semi-interpenetrating polymer network microspheres of acrylamide grafted dextran and chitosan of acyclovir. Carbohydr Polym 2007; 67(4): 605-13. [http://dx.doi.org/10.1016/j.carbpol.2006.07.001] |
| [75] | Chiappetta DA, Hocht C, Taira C, Sosnik A. Efavirenz-loaded polymeric micelles for pediatric anti-HIV pharmacotherapy with significantly higher oral bioavailability [corrected]. Nanomedicine (Lond) 2010; 5(1): 11-23. [http://dx.doi.org/10.2217/nnm.09.90] [PMID: 20025460] |
| [76] | Chiappetta DA, Hocht C, Sosnik A. A highly concentrated and taste-improved aqueous formulation of efavirenz for a more appropriate pediatric management of the anti-HIV therapy. Curr HIV Res 2010; 8(3): 223-31. [http://dx.doi.org/10.2174/157016210791111142] [PMID: 20158456] |



