- Home
- About Journals
-
Information for Authors/ReviewersEditorial Policies
Publication Fee
Publication Cycle - Process Flowchart
Online Manuscript Submission and Tracking System
Publishing Ethics and Rectitude
Authorship
Author Benefits
Reviewer Guidelines
Guest Editor Guidelines
Peer Review Workflow
Quick Track Option
Copyediting Services
Bentham Open Membership
Bentham Open Advisory Board
Archiving Policies
Fabricating and Stating False Information
Post Publication Discussions and Corrections
Editorial Management
Advertise With Us
Funding Agencies
Rate List
Kudos
General FAQs
Special Fee Waivers and Discounts
- Contact
- Help
- About Us
- Search

The Open Anatomy Journal
(Discontinued)
ISSN: 1877-6094 ― Volume 6, 2014
Morphology of Blood Cells, Liver and Spleen of the Desert Tortoise (Testudo graeca)
A Kassab*, 1, S Shousha2, A Fargani 3
Abstract
The morphological characteristics of peripheral blood cells, the anatomical structure of the liver and spleen were examined in desert tortoise. Forty healthy adult ;Testudo graeca tortoises, 20 females and 20 males were selected for this study. The tortoises were given thorough examination, and a blood sample was collected from the heart by cardiocentesis. The Wright staining method was used for classification of the blood cells. Seven different types of blood cells were determined: erythrocytes, heterophils, eosinophils, basophils, lymphocytes, monocytes and thrombocytes. Mature erythrocytes of captive Testudo graecawere nucleated ellipsoidal cells. Erythrocytes measurements for males were (length 18.76 ± 2.81 µm × width 9.52 ± 2.23 µm) and for females were (length 19.19 ± 2.90 µm × width 9.84 ± 2.50 µm). The heterophil contain large, eosinophilic, ovoid, cytoplasmic granules with eccentric nucleus. The eosinophil is distinguished by its round, eosinophilic cytoplasmic granules and the nucleus is round to oval, single or bi-lobed, and eccentrically placed within the cytoplasm. The basophil was easily identified by its deeply stained purple, large, round granules that remained tightly adhered to the centrally located nucleus. The lymphocyte contained a small amount of blue staining cytoplasm and a round nucleus with a fine reticular pattern. The monocyte contains a large amount of light blue-gray, finely granular or vacuolated cytoplasm, and an oval or indented nucleus. Thrombocytes were oval-shaped cells contained round, densely stained nucleus. The liver composed of two lobes, right and left which were connected with a narrow band of connective tissue. The parenchyma of the tortoise liver is seen to be composed of intersinusoidal cords of hepatocytes. The spleen grossly appeared as round or oval structure. The parenchyma (splenic pulp) formed of lymphoid tissue and is of two distinct types; white and red pulps.
Overall, this study provides identification of the morphological characteristics of different peripheral blood cells of the tortoise Testudo graeca species as well as the anatomy of the liver and spleen, as a reference for future hematological studies of this species and may be used as a basis for comparison in clinical cases.
Article Information
Identifiers and Pagination:
Year: 2009Volume: 1
First Page: 1
Last Page: 10
Publisher Id: TOANATJ-1-1
DOI: 10.2174/1877609400901010001
Article History:
Received Date: 26/3/2009Revision Received Date: 8/4/2009
Acceptance Date: 12/8/2009
Electronic publication date: 26/3/2009
Collection year: 2009
open-access license: This is an open access article licensed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted, non-commercial use, distribution and reproduction in any medium, provided the work is properly cited.
* Address correspondence to this author at the Department of Anatomy and Embryology, Faculty of Veterinary Medicine, Benha University, Moshtohor, 13736, Toukh, Egypt; Tel: +20133212925; 20132460640; E-mail: kassab_aa@yahoo.com
| Open Peer Review Details | |||
|---|---|---|---|
| Manuscript submitted on 26-3-2009 |
Original Manuscript | Morphology of Blood Cells, Liver and Spleen of the Desert Tortoise (Testudo graeca) | |
INTRODUCTION
The desert Tortoise is a member of the reptile family that is composed of snakes, lizards, crocodiles and other chelonians (turtles). Turtles are obviously different from other reptiles by the shell or box that completely covers the body. The shell is actually a part of the body and hardens about three years after hatching.
Tortoises have position in terrestrial live because some of population use the tortoises as source of protein food and use from zoological garden many research work studying the tortoises for fixed data about biological and behavioral and infectious diseases [1-3].
Hematological analyses have been described to a limited extent for captive desert tortoises and free-ranging tortoises in the western Mojave Desert, northeastern Mojave Desert, Sonoran Desert in Arizona, and Alachua and Lake Counties in Florida [4-1]. Understanding of blood composition of turtles is very important for preventing and treating many diseases [12].
There are many published papers that characterize the blood of land tortoise [1, 13, 14]. Data for the gopher tortoise have included red and white blood cell counts [15], erythrocyte size measurements [16], hematocrit, specific gravity, blood volume, plasma volume, and total body water [17], percent protein [18], total protein, sialic acid [19], and calcium, sodium, magnesium, and cholesterol [20].
However, little or no previous study has presented values for a wide range of blood variables for the desert tortoise Testudo graeca.
Clarification of the morphological characteristics and classification of blood cells, especially WBCs, from the desert tortoise Testudo graeca is important because this species information could be used to assist those attempting to interpret WBC counts for clinical and research specimens in the wild.
The anatomical structure of turtles alimentary canal wasn’t fully knowledged, especially morphology and asymmetry of particular organs. The one from more interesting organs of alimentary canal is the liver, which is the energy centre of the organism and blood forming organ. The macroscopic and microscopic description of this organ in turtles introduces following scientific publications [21, 22].
Therefore, the aim of this work was firstly, identification of the morphological characteristics of different peripheral blood cells of the tortoise Testudo graeca species, as a reference for future hematological studies of this species. Secondly, Description of the macro- and micro- morphology of the blood cells forming organs especially liver and spleen.
MATERIALS AND METHODS
Animals
Forty healthy adult Testudo graeca tortoises, 20 females and 20 maleswere collected during two distinct trapping seasons, fall (September-October, 2006) and spring (February-March 2007). They were collected from wildlife management area in Sidi Khalifa in Libya. Captured tortoises were physically examined and the carapace and plastron length and width and body weight were recorded. Tortoises were kept in similar plastic boxes and fed green leaves and grass. They were kept under these conditions for 21 days before the start of experiment.
Blood Sampling
Tortoises were restrained manually without sedation. Blood samples were collected by cardiocentesis. Cardiac puncture has been reported as a method of choice for blood collection. This method requires a hole drilled through the plastron on the heart site (Fig. 1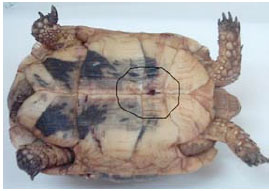 ). The 19 G luer needle was inserted in the heart. A total of one ml blood was collected into heparinized plastic containers from each experimental animal.
). The 19 G luer needle was inserted in the heart. A total of one ml blood was collected into heparinized plastic containers from each experimental animal.
 |
Fig. (1) Photograph of the adult tortoise (Testudo graeca) showing the site of cardiocentesis and Cardiac puncture through the plastron. |
Blood Cell Morphology
Blood smears were immediately prepared and air dried. Wright's stained blood smears were used for the measurement and assessment of blood cells. Slides were treated with Wright's stain for 2 minutes, washed in running tap water for 2 minutes and rinsed in distilled water for 2 minutes. Slides were dried and mounted with Canada balsam. Four to five blood smears per individual animal were randomly selected. One hundred erythrocytes and thirty of each of thrombocytes, heterophils, eosinophils, basophils, lymphocytes and monocytes were measured via Thoma’s hemocytometer by Olympus ocular micrometer (magnification power: × 1000) [16]. The measurements included maximal length (L) and maximal width (W). Sizes of cells were calculated according to the formula (L×W ×π)/4 [23, 24].
Preparation of Histological Sections
Ether was used to euthanize the animals. A saw was used to cut sides of the plastron. After removal of the plastron, the liver and spleen were examined macroscopically and specimens from the liver and spleen were obtained. They were rinsed with normal saline and then transferred into 10% neutral buffered formol solution, dehydrated and embedded in paraffin. Sections of 5-6 µm in thickness were obtained and stained with Haematoxylen and eosin (H&E) for routine histological examination [25].
The histological sections were carefully examined under an ordinary light microscope. (Carl Zeiss, Germany). Microphotography of selected microscopic fields was carried out with the aid of digital camera- computer set up.
Statistical Analysis
Numerical Data are presented as mean ± standard deviation (S.D.). Data were analyzed for statistical significance using student t test. P value of less than 0.05 indicates significance. Confidence intervals, at 95% level, were also calculated. All statistical analyses were accomplished with the statistical package for the social sciences (SPPS 9.0, SPSS Inc. Corp., Chicago, IL, USA) computer package.
RESULTS
Physical Parameters
Carapace and plastron measurements were collected from all forty adult tortoises. The mean and standard deviation for the carapace and plastron lengths and widths are reported in Table 1. Female tortoises had longer carapace length (females: 24.3 ± 1.69 cm, males 22.4 ± 1.57 cm), carapace width (females: 17.4 ± 1.63 cm, males: 14.9 ± 1.23 cm), plastron length (females: 20.4 ± 1.70 cm, males: 18.4 ± 1.53 cm), and plastron width (females: 15.5 ± 1.53 cm, males: 13.9 ± 1.29 cm) than males. Body weight means for females was (646.50 ± 18.96 g) and for males (628.90 ± 28.11 g).
Red Blood Cells Morphology
The erythrocytes of Testudo graeca (Fig. 2a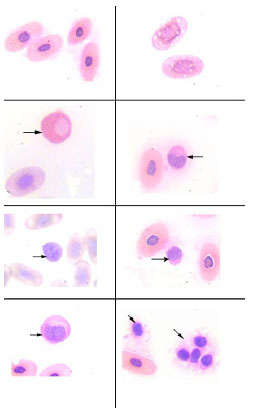 ) were nucleated, ellipsoid cells and their nuclei were oval and centrally located like those of the other reptile species. The cytoplasm of mature erythrocytes appeared light and dark pink and was homogeneous under Wright's stain. The nuclei of mature erythrocytes are chromophilic. They might contain prominent vacuoles (Fig. 2b
) were nucleated, ellipsoid cells and their nuclei were oval and centrally located like those of the other reptile species. The cytoplasm of mature erythrocytes appeared light and dark pink and was homogeneous under Wright's stain. The nuclei of mature erythrocytes are chromophilic. They might contain prominent vacuoles (Fig. 2b ). Erythrocyte measurements for males were (length 18.76 ± 2.81 µm × width 9.52 ± 2.23 µm) and for females were (length 19.19 ± 2.90 µm × width 9.84 ± 2.50 µm). The nucleus was centrally located and stained dark purple under Wright's stain. The measurements of the nucleus for males were (6.14 ± 0.81 µm × 4.44 ± 0.88 µm) and for females were (6.32 ± 0.82 µm × 4.79 ± 0.88 µm). Size means of the erythrocytes for males was (140.41 ± 49.34 µ²) and for females (142.07 ± 54.98 µ²). Results of erythrocytes measurements are summarized in Table 2.
). Erythrocyte measurements for males were (length 18.76 ± 2.81 µm × width 9.52 ± 2.23 µm) and for females were (length 19.19 ± 2.90 µm × width 9.84 ± 2.50 µm). The nucleus was centrally located and stained dark purple under Wright's stain. The measurements of the nucleus for males were (6.14 ± 0.81 µm × 4.44 ± 0.88 µm) and for females were (6.32 ± 0.82 µm × 4.79 ± 0.88 µm). Size means of the erythrocytes for males was (140.41 ± 49.34 µ²) and for females (142.07 ± 54.98 µ²). Results of erythrocytes measurements are summarized in Table 2.
White Blood Cells Morphology
Granulocytes
Granulocytes include the leukocytes that contain specific granules that identify the cell lineage. These cells include heterophils, eosinophils, and basophils.
Heterophils-The heterophil (Fig. 2c ) was the most abundant leukocyte found in blood. It is equivalent to a mammalian neutrophil. It contained large, eosinophilic and round cytoplasmic granules. The cytoplasm, which can be difficult to visualize, was light blue or clear. The nucleus was frequently displaced toward the edge of the cell and appeared basophilic with dense chromatin. Heterophil measurements in males were (length 15.53 ± 2.7 µm and width 13.49 ± 2.60 µm) and in females were (length 15.48 ± 2.52 µm and width 12.88 ± 2.54 µm). Heterphils size meansof the male was (172.72 ± 58.60 µ²) and in the female was (167.05 ± 49.24 µ²). Results of heterophils measurements are summarized in Table 3.
) was the most abundant leukocyte found in blood. It is equivalent to a mammalian neutrophil. It contained large, eosinophilic and round cytoplasmic granules. The cytoplasm, which can be difficult to visualize, was light blue or clear. The nucleus was frequently displaced toward the edge of the cell and appeared basophilic with dense chromatin. Heterophil measurements in males were (length 15.53 ± 2.7 µm and width 13.49 ± 2.60 µm) and in females were (length 15.48 ± 2.52 µm and width 12.88 ± 2.54 µm). Heterphils size meansof the male was (172.72 ± 58.60 µ²) and in the female was (167.05 ± 49.24 µ²). Results of heterophils measurements are summarized in Table 3.
Eosinophils-The eosinophil (Fig. 2d ) was distinguished by its round eosinophilic cytoplasmic granules which do not fill the colorless cytoplasm. The nucleus contains coarse, clumped chromatin and stained red. It was round to oval, single or bi-lobed and eccentrically placed within the cytoplasm. Its measurements in males were (length 14.48 ± 1.54 µm and width 10.72 ± 1.7 µm) and in females were (length 14.19 ± 1.81 µm and width 10.08 ± 1.3 µm). Eosinophil size means of the male was (122.84 ± 26.24 µ²) and in female was (118.33 ± 28.72 µ²). Results of eosinophils measurements are summarized in Table 4.
) was distinguished by its round eosinophilic cytoplasmic granules which do not fill the colorless cytoplasm. The nucleus contains coarse, clumped chromatin and stained red. It was round to oval, single or bi-lobed and eccentrically placed within the cytoplasm. Its measurements in males were (length 14.48 ± 1.54 µm and width 10.72 ± 1.7 µm) and in females were (length 14.19 ± 1.81 µm and width 10.08 ± 1.3 µm). Eosinophil size means of the male was (122.84 ± 26.24 µ²) and in female was (118.33 ± 28.72 µ²). Results of eosinophils measurements are summarized in Table 4.
Basophils-The basophil (Fig. 2e ) was easily identified by its deeply stained purple, large, round granules that remained tightly adhered to the centrally located nucleus. In some instances the granules completely occluded the nucleus. Its measurements in males were (length 12.18 ± 1.02 µm and width 11.91 ± 0.98 µm) and in females were (length 12.56 ± 1.14 µm and width 12.26 ± 1.18 µm). Basophil size means of the male was (114.89 ± 18.82 µ²) and in female was (121.99 ± 22.06 µ²). Results of basophils measurements are summarized in Table 5.
) was easily identified by its deeply stained purple, large, round granules that remained tightly adhered to the centrally located nucleus. In some instances the granules completely occluded the nucleus. Its measurements in males were (length 12.18 ± 1.02 µm and width 11.91 ± 0.98 µm) and in females were (length 12.56 ± 1.14 µm and width 12.26 ± 1.18 µm). Basophil size means of the male was (114.89 ± 18.82 µ²) and in female was (121.99 ± 22.06 µ²). Results of basophils measurements are summarized in Table 5.
Mononuclear Cells
The mononuclear cells include lymphocytes and monocytes. These cells generally lack significant lobulation of the nucleus and do not contain an abundance of cytoplasmic granules.
Lymphocytes-The lymphocytes (Fig. 2f ) contained a small amount of blue staining cytoplasm and a round nucleus with a fine reticular pattern. The lymphocyte was more spheroid than the thrombocyte, with a nuclear to cytoplasmic ratio of greater than one. Its measurements in males were (length 9.15 ± 2.17 µm and width 7.70 ± 1.77 µm) and in females were (length 10.10 ± 2.43 µm and width 8.71 ± 2.15 µm). Lymphocytes size means of the male was (58.15 ± 27.20 µ²) and in female was (73.04 ± 32.08 µ²). Results of lymphocytes measurements are summarized in Table 6.
) contained a small amount of blue staining cytoplasm and a round nucleus with a fine reticular pattern. The lymphocyte was more spheroid than the thrombocyte, with a nuclear to cytoplasmic ratio of greater than one. Its measurements in males were (length 9.15 ± 2.17 µm and width 7.70 ± 1.77 µm) and in females were (length 10.10 ± 2.43 µm and width 8.71 ± 2.15 µm). Lymphocytes size means of the male was (58.15 ± 27.20 µ²) and in female was (73.04 ± 32.08 µ²). Results of lymphocytes measurements are summarized in Table 6.
Monocytes-The monocyte (Fig. 2g) contained a large amount of light blue-gray, finely granular or vacuolated cytoplasm and an oval or indented nucleus with a dense chromatin pattern. Monocytes in males were (length 14.67 ± 2.53 µm and width 10.28 ± 2.05 µm) and in females were (length14.99 ± 2.83 µm and width 10.91 ± 1.93). Size means of the monocyte in male was (118.32 ± 49.34 µ²) and in female was (125.74 ± 59.97 µ²). Results of monocytes measurements are summarized in Table 7.
Thrombocytes - The thrombocytes (Fig. 2h) were ellipsoidal cells that contained round densely stained nucleus. The cytoplasm was clear and the thrombocytes tended to clump together. These latter two identifying characteristics help in distinguishing the thrombocytes from the lymphocytes. The thrombocytes in males were (length 6.18 ± 0.92 x width 4.59 ± 0.95) and in females were (length 6.29 ± 1.00 and width 4.62 ± 1.02). Thrombocyte size means of the male was (22.70 ± 7.14 µ²) and in female was (23.21 ± 7.59 µ²). Results of thrombocytes measurements are summarized in Table 8.
Morphological and Histological Results of the Liver
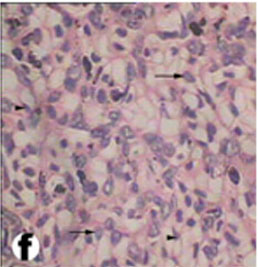
The liver of tortoise is situated in the abdominal cavity transverse to the long axis of the body. It is dark red in the fresh condition. It was composed of two lobes, right and left (Fig. 3). The left lobe is slight larger than the right one. The two lobes were connected with a narrow band of connective tissue. In a section examined under low power, the parenchyma of the tortoise liver is seen to be composed of intersinusoidal cords of hepatocytes (Figs. 4 and 5). The parenchymal cells (hepatocytes) arranged as cords of cells with vascular sinusoid spaces between the cords. The cords radiate from central vein which are the centers of the classic hepatic lobules (Fig. 4). Hepatic cells were polygonal and of different sizes and their nuclei were spherical or ovoid (Figs. 5 and 6). The portal areas (hepatic triad) present and comprise branches of portal vein, hepatic artery and bile duct, often also with a lymphatic vessel, lying in a small amount of connective tissue (Figs. 6 and 7). Within the lobule, there is a fine meshwork of reticular and collagenous fibers around the sinusoids and within the perisinusoidal spaces (Fig. 8).
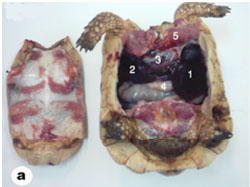 |
Fig. (3) Photograph showing the Viscera of the tortoise. 1- Left lobe of liver, 2- Right lobe of liver, 3- Heart, 4- Stomach, 5- Pectoralis muscles. |
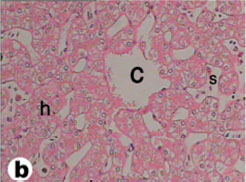 |
Fig. (4) Light micrograph of liver tissue from tortoise. c- Central vein, h- sheets of hepatocytes, s- Blood sinusoid containing red blood cells. (H & E 400) |
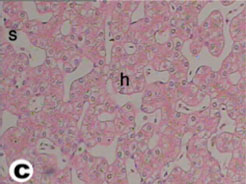 |
Fig. (5) Light micrograph of liver tissue from tortoise showing cords of hepatocytes (h) with their nuclei and the blood sinusoids(s). (H & E 400). |
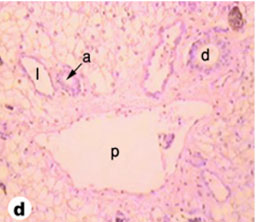 |
Fig. (6) Light micrograph of tortoise liver showing the portal area with a branch of the portal vein (p), a branch of the hepatic artery (a), a bile duct (b) and a lymphatic vessel (l). (H & E 160). |
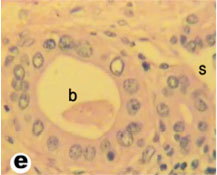 |
Fig. (7) High magnification of light micrograph of tortoise liver showing the bile duct (b) and sinusoids (s). (H & E 800). |
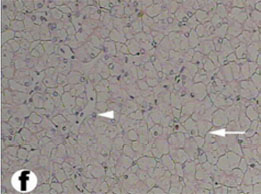 |
Fig. (8) High magnification of Light micrograph of the liver parenchyma showing a typical reticulum (arrow) and hepatocytes (arrow heads). (H & E 400). |
Morphological and Histological Results of the Spleen
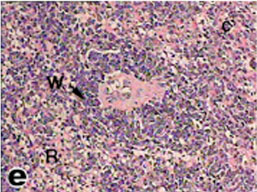
The spleen of tortoise is the most important immune organ and it is the body's largest filter of the blood. The spleen appeared grossly as round or oval structure. It was located in the abdomen of tortoise under the stomach (Fig. 9). Sections of tortoise spleen showed normal splenic structure. The organ was surrounded by a fibrous capsule of connective tissue, stemming from which are trabeculae that support the large vasculature (Fig. 10). The connective tissue capsule and trabeculae extending from it were clearly identified. The parenchyma (splenic pulp) formed of lymphoid tissue and is of two distinct types; white and red pulps (Fig. 11). The white pulp composed of central arteriole surrounded with aggregation of lymphoid tissues (Figs. 12, 13 and 16). The red pulp is more abundant and formed of vascular sinuses filled with blood cells (Figs. 12, 14 and 15).
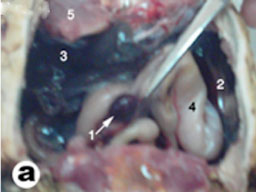 |
Fig. (9) Topography of the liver and spleen of tortoise. 1- spleen, 2- left lobe of liver, 3- Right lobe of liver, 4- stomach, 5- Pectoralis muscles. |
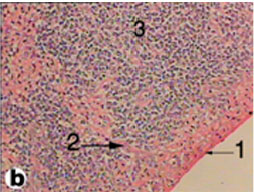 |
Fig. (10) Light micrograph of spleen tissue from tortoise. 1- Capsule, 2- Trabecula, 3- Parenchyma. (H & E 200) |
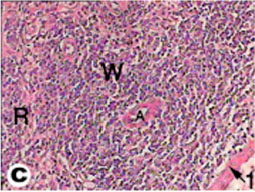 |
Fig. (11) Light micrograph of spleen tissue from tortoise. 1- Capsule, A- Central arteriole, W- White pulp, R- Red pulp. (H & E 400). |
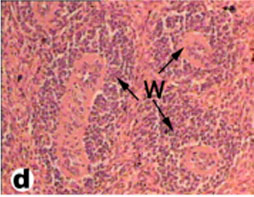 |
Fig. (12) Light micrograph of spleen tissue from tortoise showing three white pulps (W) in the splenic parenchyma. (H & E 400). |
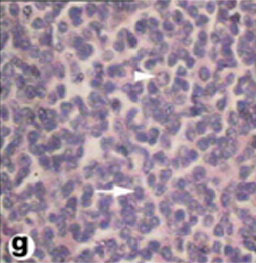 |
Fig. (15) High magnification of Light micrograph of white pulp of the spleen, showing lymphocytes (arrow) and plasma cell (arrow head). (H & E 800). |
DISCUSSION
This study represents the first attempt to develop baseline health parameters for the desert tortoise Testudo graeca. The original goal for this study was to study the morphological characteristics of the blood cells and the anatomy of the liver and spleen. The data presented will provide the veterinary clinician with specific hematological data which will allow for a more comprehensive medical assessment in tortoise clinical cases.
Our findings related to carapace and plastron measurements suggest that there is minimal variation in the size of the tortoises. It was interesting to note that the female tortoises were larger and heavier than the male tortoises in this study. Diaz-Figueroa [26] and McRae et al. [27] reported the same observations with gopher tortoise. In general, male tortoises are larger than female tortoises [28, 29]. This difference may be the result of older and larger male tortoises being removed from the population.
Hematology and blood biochemical parameters are essential to assess the health and physiological status of stoic animals like the gopher tortoise. Although one hundred-sixty blood samples are routinely collected from tortoises to perform complete blood counts, morphologic criteria for distinguishing the various circulating blood cells have not been established in the desert tortoise Testudo graeca. Alleman et al. [13] described the morphological features and cytochemical staining characteristics of circulating blood cells for the desert tortoise. However, these same features also need to be described for the desert tortoise Testudo graeca. The hematological parameters measured in this study were considered to be within reference limits for captive chelonian species. The findings suggest that these tortoises are, in general, healthy. The complete blood counts in tortoises captured during the fall were significantly lower than in the spring. The same seasonal variation was noted in gopher tortoises from Florida [6]. Lower total white blood cell counts are not an uncommon finding in reptiles during the fall. As the environmental temperature decreases, the reptile’s metabolic rate and immune function also decrease. Lymphocytes were the predominant white cell found in these tortoises. The lymphocytes play several important roles in the reptile, including producing antibodies and attacking foreign materials (natural killer cells).
Erythrocytes are morphologically similar among various species of reptiles [30]. The mature erythrocytes were nucleated ellipsoidal cells with pink stained cytoplasm, which are consistent with that reported in desert tortoise [13], in European pond turtles [24] and in green turtles [31]. The erythrocyte measurements are also in agreement with the results of Colagar and Jafari [32] and Ugˇurtas et al. [23].
Some erythrocytes in the desert tortoise Testudo graeca had small intracytoplasmic inclusions. Similar round or irrigular intracytoplasmic inclusions have been described in the erythrocytes of several species of reptiles [33], in water dragon [34], in desert tortoise [13], in Juvenile Loggerhead Sea turtle [35] and in green sea turtles [36]. These intracytoplasmic inclusions were considered ultrastructurally to represent a degenerate organelle, possibly mitochondria [13]. In mammals, the nuclear lobulation is distinct, but this is not a prominent feature of turtle granulocytes.
Saint Girons [30] reported the presence of eosinophils, azurophils, neutrophils and plasma cells in reptiles. While, Sypek and Borysenko [37] reported eosinophils, heterophils, basophils, monocytes and lymphocytes. According to Canfield [38], the mammalian neutrophil is equivalent to the non mammalian heterophil. The morphological characteristics obtained by light microscopy of erythrocytes, thrombocytes, monocytes and lymphocytes were similar to those seen in other reptilian species [13, 36, 39].
The heterophil contains large, eosinophilic, ovoid, cytoplasmic granules with eccentric nucleus. These features are similar to those described for heterophils from Loggerhead Sea turtle [35] and green sea turtles [36]. However, other reptiles, particularly the snakes, yellow rat snake ( Elaphe obsolete quadrivittata), eastern diamond back rattle snake (Crotalus adamanteus) and the king cobra (Ophiophagus hannah), had two morphological variations of heterophils by electron microscope; one variant contained a homogenous population of electon-dense intracytoplasmic granules and the other variant contained some granules that were electron dense whereas other granules similar in size and shape, suggesting different developmental stages of heterophils in the circulation [40, 41].
The eosinophil is distinguished by its round, eosinophilic cytoplasmic granules and the nucleus is round to oval, single or bi-lobed, and eccentrically placed within the cytoplasm. Eosinophils from desert tortoise were homogenous in size as in Loggerhead turtles [35], unlike eosinophils from green turtles [36] and Kemp's ridley turtles [42], which are large as well as small. Work et al. [36] suspected that the large eosinophils in green turtles represent activated cells in response to a parasitic infestation or other inflammatory stimulus.
The lymphocyte contains a small amount of blue staining cytoplasm and a round nucleus with a fine reticular pattern. These characteristics are similar to those described for heterophils from loggerhead sea turtle [35] and green sea turtles [36].
The monocyte contains a large amount of light blue-gray, finely granular or vacuolated cytoplasm, and an oval or indented nucleus. These characteristics are similar to those described for heterophils from Loggerhead Sea turtle [35] and green sea turtles [36]. However, other authors didn't identify monocyte in the blood of green turtles [43,44] or Kemp's ridley turtles [42].
The thrombocytes were oval-shaped cells contain round, densely staining nucleus, although sometimes were observed round thrombocytes, similar to those seen in Loggerhead Sea turtle [35] and green sea turtles [36].
Concerning the morphology of the liver of the desert tortoise Testudo graeca, the results of the present workhave many similarities to that described by Marycz and Rogowska [22] in Horsefield's (Testudo horsefieldi) and Hermann's tortoises (Testudo hermanni). Generally, within Testudinata, the liver had very similar shape and morphology. The differences most often concerned with the mass of this organ, which at carnivorous tortoises was relatively greater [45]. While this organ could appeared as long single lobe and spindle - shaped organ at snakes, or the multilobe form at other reptiles [21].
Concerning the morphology of the spleen of the desert tortoise Testudo graeca, our results revealed that it has red and white pulps that come in agreement with that mentioned by Borysenko [46], in the snapping turtle (Chelydra serpentine).
The liver and spleen of tortoise differs from that of mammals and similar to reptiles which may contribute the understanding of evolutionary requirements of the species.
In conclusion; this study is the first attempt to characterize the health status of the desert tortoise Testudo graeca. In general, our findings suggest that these tortoises are in good health. However, some of our findings suggest that these keystone species should continue to be monitored to protect them into the future.