- Home
- About Journals
-
Information for Authors/ReviewersEditorial Policies
Publication Fee
Publication Cycle - Process Flowchart
Online Manuscript Submission and Tracking System
Publishing Ethics and Rectitude
Authorship
Author Benefits
Reviewer Guidelines
Guest Editor Guidelines
Peer Review Workflow
Quick Track Option
Copyediting Services
Bentham Open Membership
Bentham Open Advisory Board
Archiving Policies
Fabricating and Stating False Information
Post Publication Discussions and Corrections
Editorial Management
Advertise With Us
Funding Agencies
Rate List
Kudos
General FAQs
Special Fee Waivers and Discounts
- Contact
- Help
- About Us
- Search

The Open Bioactive Compounds Journal
(Discontinued)
ISSN: 1874-8473 ― Volume 9, 2020
Harpin Proteins Improve Bioactive Compounds Content in Crimson Seedless Table Grape
Pasquale Crupi1, *, Giambattista Debiase1, Gianvito Masi1, Francesca Mangione2, Luigi Tarricone1
Abstract
Background:
Frequently, in warm climates such as Mediterranean areas, a red-pink table grape ‘Crimson Seedless’ does not reach a good berry skin color; and an acceptable anthocyanin bioactive compounds content, responsible for the red color of berries. Harpin proteins are biotechnologically developed bio-activators that, if applied on plants during the growing period, trigger the expression of hundreds of genes among which those associated with the biosynthesis of bioactive compounds (such as anthocyanins).
Aim:
This research aimed at using harpin proteins to test their suitability in improving the grape skin color.
Methods and Materials:
Beta-harpin protein 1% p.a. (400 g/Ha) was applied to ‘Crimson Seedless’ vines three times at the beginning of veraison. Six samplings were carried out for both the treated and control grapes until commercial harvest. In the skin extracts, total and individual anthocyanins content was determined by UV-Vis and RP-HPLC-DAD analyses, respectively.
Results:
The collected results confirmed that the application of harpin proteins effectively stimulated the anthocyanin biosynthesis leading to make peonidin-3O-glucoside, cyanidin-3O-glucoside, and malvidin-3O-glucoside values from 2 to almost 10 folds higher in treated grapes than in control grapes (P < 0.05).
Conclusion:
Actually, harpin proteins improved the color of the berry skin, leading to a significantly higher concentration of anthocyanins in treated than in control grapes.
Article Information
Identifiers and Pagination:
Year: 2019Volume: 7
First Page: 1
Last Page: 7
Publisher Id: TOBCJ-7-1
DOI: 10.2174/1874847301907010001
Article History:
Received Date: 29/01/2019Revision Received Date: 09/04/2019
Acceptance Date: 22/04/2019
Electronic publication date: 31/05/2019
Collection year: 2019
open-access license: This is an open access article distributed under the terms of the Creative Commons Attribution 4.0 International Public License (CC-BY 4.0), a copy of which is available at: (https://creativecommons.org/licenses/by/4.0/legalcode). This license permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
* Address correspondence to this author at the CREA-VE - Council for Agricultural Research and Economics – Research Centre for Viticulture and Enology, Via Casamassima 148 - 70010 Turi (BA) – Italy; Tel: +390808915711; Fax: +390804512925; E-mail: pasquale.crupi@crea.gov.it
| Open Peer Review Details | |||
|---|---|---|---|
| Manuscript submitted on 29-01-2019 |
Original Manuscript | Harpin Proteins Improve Bioactive Compounds Content in Crimson Seedless Table Grape | |
1. INTRODUCTION
Anthocyanins are the pigments which determine the color of grape varieties. Grapes with high levels of these compounds in their skin appear darker and more red-colored than grapes with low levels of anthocyanins, even though the relationship between pigments content and berry color is not linear [1Peppi MC, Fidelibus MW, Dokoozliam N. Application timing and concentration of abscisic acid affect the quality of ‘Redglobe’ grapes. J Hortic Sci Biotechnol 2007; 82: 304-10.
[http://dx.doi.org/10.1080/14620316.2007.115122 33] , 2Lurie S, Ovadia R, Nissim-Levi A, et al. Abscisic acid improves colour development in ‘Crimson Seedless’ grapes in the vineyard and on detached berries. J Hortic Sci Biotechnol 2009; 84: 639-44.
[http://dx.doi.org/10.1080/14620316.2009.11512579] ]. The structure of anthocyanin consists of a benzopyrilium moiety substituted by different groups (mainly methoxyls and/or hydroxyls), whose number and position give rise to delphinidin, cyanidin, petunidin, peonidin, and malvidin; being chemically more stable, they are present in the grapes’ skin as 3-O-glucosides derivatives [3Guidoni S, Hunter JJ. Anthocyanin profile in berry skins and fermenting must/wine, as affected by grape ripeness level of Vitis vinifera cv. Shiraz/R99. Eur Food Res Technol 2012; 235: 397-408.
[http://dx.doi.org/10.1007/s00217-012-1744-5] ]. Besides having a key role for the commercial quality of grapes, nowadays, there is a growing interest in anthocyanins as bioactive compounds, principally due to their biological properties, including but not limited to antioxidant, anti-inflammatory, anticancer, and cardioprotective [4Carrieri C, Milella RA, Incampo F, et al. Antithrombotic activity of 12 table grape varieties. Relationship with polyphenolic profile. Food Chem 2013; 140(4): 647-53.
[http://dx.doi.org/10.1016/j.foodchem.2012.10.132] [PMID: 236927 48] -7Đorđević NO, Todorović N, Novaković IT, et al. Antioxidant activity of selected polyphenolics in yeast cells: The case study of Montenegrin Merlot Wine. Molecules 2018; 23(8): 1971.
[http://dx.doi.org/10.3390/molecules23081971] [PMID: 300872 28] ].
Anthocyanins are the pigments which determine the color of grape varieties. Grapes with high levels of these compounds in their skin appear darker and more red-colored than grapes with low levels of anthocyanins, even though the relationship between pigments content and berry color is not linear [1Peppi MC, Fidelibus MW, Dokoozliam N. Application timing and concentration of abscisic acid affect the quality of ‘Redglobe’ grapes. J Hortic Sci Biotechnol 2007; 82: 304-10.
[http://dx.doi.org/10.1080/14620316.2007.115122 33] , 2Lurie S, Ovadia R, Nissim-Levi A, et al. Abscisic acid improves colour development in ‘Crimson Seedless’ grapes in the vineyard and on detached berries. J Hortic Sci Biotechnol 2009; 84: 639-44.
[http://dx.doi.org/10.1080/14620316.2009.11512579] ]. The structure of anthocyanin consists of a benzopyrilium moiety substituted by different groups (mainly methoxyls and/or hydroxyls), whose number and position give rise to delphinidin, cyanidin, petunidin, peonidin, and malvidin; being chemically more stable, they are present in the grapes’ skin as 3-O-glucosides derivatives [3Guidoni S, Hunter JJ. Anthocyanin profile in berry skins and fermenting must/wine, as affected by grape ripeness level of Vitis vinifera cv. Shiraz/R99. Eur Food Res Technol 2012; 235: 397-408.
[http://dx.doi.org/10.1007/s00217-012-1744-5] ]. Besides having a key role for the commercial quality of grapes, nowadays, there is a growing interest in anthocyanins as bioactive compounds, principally due to their biological properties, including but not limited to antioxidant, anti-inflammatory, anticancer, and cardioprotective [4Carrieri C, Milella RA, Incampo F, et al. Antithrombotic activity of 12 table grape varieties. Relationship with polyphenolic profile. Food Chem 2013; 140(4): 647-53.
[http://dx.doi.org/10.1016/j.foodchem.2012.10.132] [PMID: 236927 48] -7Đorđević NO, Todorović N, Novaković IT, et al. Antioxidant activity of selected polyphenolics in yeast cells: The case study of Montenegrin Merlot Wine. Molecules 2018; 23(8): 1971.
[http://dx.doi.org/10.3390/molecules23081971] [PMID: 300872 28] ].
Anthocyanin biosynthesis and accumulation in the skin cells start from veraison (the onset of maturation) until the harvest and are mainly under genetic control [8Gagné S, Cluzet S, Mérillon JM, Gény L. ABA initiates anthocyanin production in grape cell cultures. J Plant Growth Regul 2011; 30: 1-10.
[http://dx.doi.org/10.1007/s00344-010-9165-9] ]; however, climatic conditions and cultural practices, including the use of exogenous Plant Growth Regulators (PGRs), frequently influence the gene expression and activation of the biosynthetic enzymes [2Lurie S, Ovadia R, Nissim-Levi A, et al. Abscisic acid improves colour development in ‘Crimson Seedless’ grapes in the vineyard and on detached berries. J Hortic Sci Biotechnol 2009; 84: 639-44.
[http://dx.doi.org/10.1080/14620316.2009.11512579] , 9Ferrara G, Mazzeo A, Matarrese AMS, et al. Ethephon as a potential abscission agent for table grapes: Effect on pre-harvest abscission, fruit quality, and residue. Front Plant Sci 2016; 7: 620.
[http://dx.doi.org/10.3389/fpls.2016.00620] [PMID: 273034 07] -11Vujović D, Maletić R, Popović-Đorđević J, Pejin B, Ristić R. Viticultural and chemical characteristics of Muscat Hamburg preselected clones grown for table grapes. J Sci Food Agric 2017; 97(2): 587-94.
[http://dx.doi.org/10.1002/jsfa.7769] [PMID: 270982 41] ]. In this context, some varieties (i.e. ‘Crimson Seedless’) are known to fail in achieving the desired level of red color at harvesting, especially in very warm climates, maybe due to consistently high temperature in summer together with the narrow day/night temperature range [12Spayd SE, Tarara JM, Mee DL, Ferguson JC. Separation of sunlight and temperature effects on the composition of Vitis vinifera cv. Merlot berries. Am J Enol Vitic 2008; 53: 171-82., 13Crupi P, Bergamini C, Perniola R, Dipalmo T, Clodoveo ML, Antonacci D. A chemometric approach to identify the grape cultivar employed to pruduce nutraceutical fruit juice. Eur Food Res Technol 2015; 241: 487-96.
[http://dx.doi.org/10.1007/s00217-015-2478-y] ].
Harpin Proteins (HrP), encoded by hpr (hypersensitive response and pathogenicity) genes from gram-negative plant pathogenic bacteria, are non-host-specific elicitors of the hypersensitive response [14Alfano JR, Collmer A. The type III (Hrp) secretion pathway of plant pathogenic bacteria: Trafficking harpins, Avr proteins, and death. J Bacteriol 1997; 179(18): 5655-62.
[http://dx.doi.org/10.1128/jb.179.18.5655-5662.1997] [PMID: 92944 18] ] and can be considered as PGR, in the sense that, if applied on plants during the growing period, they trigger the expression of hundreds of genes related to the plant disease resistance, among which those involved in the biosynthesis of bioactive compounds (i.e. VvPAL, VvCHS, and VvUFGT) such as anthocyanins [15Li M, Yu ML, Zhang ZQ, Liu QN, Wu YC, Liu ZG. Effects of Harpin protein on fruit quality and shelf life of winter jujube (Ziziphus jujube Mill. Cv. Dongzao). Biol Agric Hortic 2013; 29: 58-68.
[http://dx.doi.org/10.1080/01448765.2012.762729] -17Akbuk N, Tezcan H, Akbudak B, Seniz V. The effect of harpin protein on plant growth parameters, leaf chlorophyll, leaf colour and percentage rotten fruit of pepper plants inoculated with Botrytis cinerea. Sci Hortic (Amsterdam) 2006; 109: 107-12.
[http://dx.doi.org/10.1016/j.scienta.2006.03.008] ]. Thus, even though to the best of our knowledge, no reports exist about HrP application on grapes for improving berry skin color, it could be hypothesized that their use is effective in triggering the anthocyanins biosynthesis and enhancing their content in grape skin, too.
Therefore, the aim of this work was to apply foliar spraying on ‘Crimson Seedless’ grapes, a HrP commercial formulation (named PhCO2 – SIPCAM Italia S.p.A.), in order to improve the color and the anthocyanin content in grapes’ skin for overcoming the issue related to the coloration defect and giving higher nutraceutical quality to this variety.
2. EXPERIMENTAL
2.1. Plant Materials
The experiment was conducted in 2017 on a commercial vineyard of ‘Crimson Seedless’ table grape (Vitis vinifera L.), grafted onto 1103 Paulsen rootstock, located in the countryside of Pulsano, Southern Italy (40°22'3” N; 17°22'46” E, 21 m a.s.l.). Vines were spaced 2.5 x 2.5 m, and trained to an overhead trellis system ‘tendone’. The growing techniques were performed according to the viticultural practices for table grapes; moreover, at the beginning of veraison, 400 g/Ha of PhCO2 was applied by foliar spraying three times (from 7th July to 1st August 2017). A randomized block design was employed, consisting of four rows of 30 vines divided into two sections and each section was treated with PhCO2 (T2) or untreated as control (T1). Finally, 4 replicates for all sections were obtained.
In the period between 81 and 89 BBCH [18Lorenz DH, Eichhorn KW, Blei-holder H, Klose R, Meier U, Weber E. Phänologische Entwicklungsstadien der Weinrebe (Vitis vinifera L.ssp. vinifera). Vitic Enol Sci 1994; 49: 66-70.] stage, from veraison to harvest, 6 samplings of 3 bunches from each replicate were carried out; besides, 10 berries from a bunch were homogenously removed (from the top, middle, and bottom of the bunch), with their pedicel still attached, and stored at -20 °C until anthocyanins’ extraction. Then, other samples of 60 berries (20 per bunch) were collected for chemical and color analyses .
2.2. Chemicals
Formic acid, acetonitrile and water HPLC grade were purchased from J. T. Baker (Deventer, Holland). Ethanol and hydrochloric acid were purchased from Carlo Erba (Milano, Italy). Sodium hydroxide (NaOH) 0.1N and bromothymol blue were purchased from Sigma Aldrich (Milano, Italy). Delphinidin-3-O-glucoside, cyanidin-3-O-glucoside, petunidin-3-O-glucoside, peonidin-3-O-glucoside, and malvidin-3-O-glucoside chlorides were purchased from Extrasynthese (Genay, France) and used as HPLC and UV-Vis spectrophotometer reference standards.
2.3. Chemical and Color Analyses on Grapes
Total Soluble Solids (TSS), Titratable Acidity (TA), and pH were determined according to the protocols established by the OIV [19Office International de la Vigne et du Vin. 1990.]. Berries were crushed to determine TSS (expressed as g/L) of berry juice using a portable refractometer (ATAGO PR32). Even TA (as g/L of tartaric acid equivalents) was determined on the juice by titrating with 0.1 N sodium hydroxide to the bromothymol blue endpoint. Finally, juice pH was measured too by means of a pH meter CRISON BASIC 20. Regarding the color of the berries, it was determined through a chroma meter CM-5 (Konica Minolta, Chiyoda, Tokyo, Japan) using the Commission Internationale de l’Eclairage Lab (CIELAB) color system, evaluating lightness, L* (0, black – 100, white), chroma, C* (0, achromatic), and hue angle on the color wheel, h (0, red – 90, yellow – 180, green – 270, blue), as previously described [20Ferrara G, Mazzeo A, Matarrese AMS, et al. Application of abscisic acid (S-ABA) to ‘Crimson Seedless’ grape berries in a Mediterranean climate: Effects on color, chemical characteristics, metabolic profile, and S-ABA concentration. J Plant Growth Regul 2013; 32: 491-505.
[http://dx.doi.org/10.1007/s003 44- 012- 9316-2] ].
2.4. Extraction of Anthocyanins from Grapes
From the frozen 10 berry samples, the skins were manually separated and cleaned by the pulp; they were dried at 30 °C for 24h, grounded and then ~ 0.4 g of powder was extracted by water/ethanol/hydrochloric acid 30:70:1 at 0.07 (g/mL) skins/extraction solvent ratio. The extraction procedure was carried out in an ultrasonic bath (SONICA 2200 EP, SOLTEC, Milano, Italy); the mixtures, placed in poly-propylene tube (15 mL), were sonicated for 21 min at a controlled temperature 50 °C. All the extracts were centrifuged at 4000g for 3 min, filtered through a 0.45 μm syringe cellulose filter, and analyzed by UV-Vis and HPLC-DAD.
2.5. UV-Vis and HPLC-DAD Analyses
The total monomeric anthocyanin pigment content was determined as proposed by Vujovic et al. 2016 [21Vujović D, Pejin B, Popović Djordjević J, Velicković M, Tesević V. Phenolic natural products of the wines obtained from three new Merlot clone candidates. Nat Prod Res 2016; 30(8): 987-90.
[http://dx.doi.org/10.1080/14786419.2015.1079191] [PMID: 263085 89] ]. HPLC-DAD analyses were performed by HPLC 1100 (Agilent Technologies, Palo Alto, USA) equipped with a degasser, a quaternary pump for solvent delivery, a thermostat column compartment, and a diode array detector. The reversed stationary phase employed was a Zorbax SB C18 5 µm (250 x 4.6 mm i.d., Agilent) with a pre-column Gemini C18 5 µm (4 x 2 mm i.d., Phenomenex). The used gradient system with acetonitrile (solvent A) and water/formic acid (90:10 v/v) (solvent B) is shown in Fig. (S1 ) with termination time 65 min. The flow was maintained at 0.7 mL/min, the column temperature at 22 °C; and the sample injection was of 5 µL. Diode array detection was between 220 and 700 nm, and absorbance was recorded at 520 nm. Anthocyanins were tentatively identified (Fig. S2
) with termination time 65 min. The flow was maintained at 0.7 mL/min, the column temperature at 22 °C; and the sample injection was of 5 µL. Diode array detection was between 220 and 700 nm, and absorbance was recorded at 520 nm. Anthocyanins were tentatively identified (Fig. S2 ) by matching the elution pattern with those of the reference standards and data reported in the literature [13Crupi P, Bergamini C, Perniola R, Dipalmo T, Clodoveo ML, Antonacci D. A chemometric approach to identify the grape cultivar employed to pruduce nutraceutical fruit juice. Eur Food Res Technol 2015; 241: 487-96.
) by matching the elution pattern with those of the reference standards and data reported in the literature [13Crupi P, Bergamini C, Perniola R, Dipalmo T, Clodoveo ML, Antonacci D. A chemometric approach to identify the grape cultivar employed to pruduce nutraceutical fruit juice. Eur Food Res Technol 2015; 241: 487-96.
[http://dx.doi.org/10.1007/s00217-015-2478-y] , 22Ferrara G, Mazzeo A, Matarrese AMS, et al. Application of abscisic acid (S-ABA) and sucrose to improve colour, anthocyanin content and antioxidant activity of cv. Crimson Seedless grape berries. Aust J Grape Wine Res 2015; 21: 18-29.
[http://dx.doi.org/10.1111/ ajgw.1211 2] ]; then, they were quantified according to the external standard method with a calibration curve obtained by the injection of 5 standard solutions at different concentrations (ranging from 50 to 300 μg/mL) of cyanidin-3-O-glucoside (R2 = 0.9996). Results were expressed as μg cyanidin-3-O-glucoside equivalents per g of dry skins.
2.6. Statistical Analysis
All the collected data were analyzed by STATISTICA 8.0 (StatSoft Inc., Tulxa, OK) software package. Specifically, after testing their normal distribution by Shapiro-Wilk’s W test together with their homoscedasticity by means of the Levene test, a two-way Analysis of Variance (ANOVA) followed by a Tukey HSD post hoc test was performed for the identified anthocyanins. The effects of ripening and harpin proteins treatment and their interactions were evaluated (Table 1). Only the significant interactions, regarding the total anthocyanins obtained by UV-Vis spectrophotometric analyses, are shown in bar graphs (Fig. 1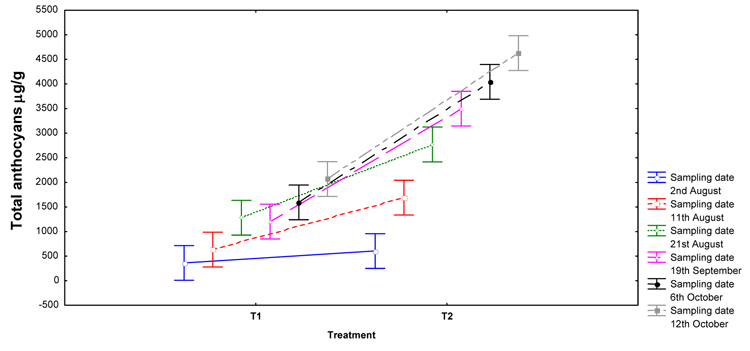 ). Instead, an independent t-test for the mean differences was applied to L*, C*, and h color parameters, graphically discussed by Box & Whisker plots (Fig. 4
). Instead, an independent t-test for the mean differences was applied to L*, C*, and h color parameters, graphically discussed by Box & Whisker plots (Fig. 4 ). Finally, histogram graphics were constructed by using Microsoft Office Excel 2007 (Figs. 2
). Finally, histogram graphics were constructed by using Microsoft Office Excel 2007 (Figs. 2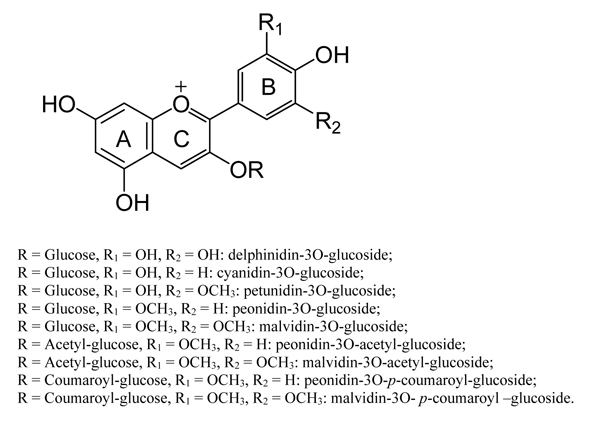 and 3
and 3 ).
).
 |
Fig. (2) Structures of the 9 anthocyanins detected in the ‘Crimson Seedless’ skin extracts. |
3. RESULTS AND DISCUSSION
The evolution of the total anthocyanins (expressed as μg/g) extracted from ‘Crimson Seedless’ grape skins during the maturation period (2nd August – 12nd October) and as affected by PhCO2 treatment is depicted in Fig. (1 ). Both the two factors “treatment” and “ripening” were statistically significant (p < 0.001; F = 289.86 and 76.62, respectively), as well as their interaction (p < 0.001; F = 14.47). It showed that the concentration of anthocyanins increased from veraison to harvest, as expected from the increasing maturation index (TSS/TA; Table 1), and more intensely in grapes treated with harpin proteins (T2). The accumulation frequency of these compounds conformed to what had been observed in Crimson during the ripening period, with an increment from veraison until, approximately, 30 days after veraison, followed by a stable level until harvest [23Singh Brar H, Singh Z, Swinny E. Dynamics of anthocyanin and flavonol profiles in the ‘Crimson Seedless’ grape berry skin during development and ripening. Sci Hortic (Amsterdam) 2008; 117: 349-56.
). Both the two factors “treatment” and “ripening” were statistically significant (p < 0.001; F = 289.86 and 76.62, respectively), as well as their interaction (p < 0.001; F = 14.47). It showed that the concentration of anthocyanins increased from veraison to harvest, as expected from the increasing maturation index (TSS/TA; Table 1), and more intensely in grapes treated with harpin proteins (T2). The accumulation frequency of these compounds conformed to what had been observed in Crimson during the ripening period, with an increment from veraison until, approximately, 30 days after veraison, followed by a stable level until harvest [23Singh Brar H, Singh Z, Swinny E. Dynamics of anthocyanin and flavonol profiles in the ‘Crimson Seedless’ grape berry skin during development and ripening. Sci Hortic (Amsterdam) 2008; 117: 349-56.
[http://dx.doi.org/10.1016/j.scienta.2008.05.007] ].
Thereby, PhCO2 treatment strongly affected the color development in the grape skin, indeed the level of anthocyanins at the harvest time was more than twice to that of the control fruit (2100 vs 4600 μg/g of dry skins in T1 and T2, respectively); even though the significant influence of the treatment was just observed from the second sampling date (Fig. 1 ). This finding was in agreement with previous researches, which demonstrated the positive effect of exogenous applied PGRs (such as S-ABA and ethephon), favoring the biosynthesis of pigments and overall improving the berry color in Crimson Seedless grapes [2Lurie S, Ovadia R, Nissim-Levi A, et al. Abscisic acid improves colour development in ‘Crimson Seedless’ grapes in the vineyard and on detached berries. J Hortic Sci Biotechnol 2009; 84: 639-44.
). This finding was in agreement with previous researches, which demonstrated the positive effect of exogenous applied PGRs (such as S-ABA and ethephon), favoring the biosynthesis of pigments and overall improving the berry color in Crimson Seedless grapes [2Lurie S, Ovadia R, Nissim-Levi A, et al. Abscisic acid improves colour development in ‘Crimson Seedless’ grapes in the vineyard and on detached berries. J Hortic Sci Biotechnol 2009; 84: 639-44.
[http://dx.doi.org/10.1080/14620316.2009.11512579] , 9Ferrara G, Mazzeo A, Matarrese AMS, et al. Ethephon as a potential abscission agent for table grapes: Effect on pre-harvest abscission, fruit quality, and residue. Front Plant Sci 2016; 7: 620.
[http://dx.doi.org/10.3389/fpls.2016.00620] [PMID: 273034 07] , 22Ferrara G, Mazzeo A, Matarrese AMS, et al. Application of abscisic acid (S-ABA) and sucrose to improve colour, anthocyanin content and antioxidant activity of cv. Crimson Seedless grape berries. Aust J Grape Wine Res 2015; 21: 18-29.
[http://dx.doi.org/10.1111/ ajgw.1211 2] ].
The mean values of the pigments identified and quantified (by HPLC-DAD analyses) in ‘Crimson Seedless’ skins, as affected by the two experimental factors, are reported in Table 1. The effect of harpin proteins on the main anthocyanins singularly was also assessed. In accordance with the literature, the cultivar was prevalently characterized by dihydroxylated anthocyanins, in particular, peonidin-3O-glucoside (Fig. 2 ), as typically found in pink and red colored table grape varieties [10Crupi P, Antonacci D, Savino M, Genghi R, Perniola R, Coletta A. Girdling and gibberellic acid effects on yield and quality of a seedless red table grape for saving irrigation water supply. Eur J Agron 2016; 80: 21-31.
), as typically found in pink and red colored table grape varieties [10Crupi P, Antonacci D, Savino M, Genghi R, Perniola R, Coletta A. Girdling and gibberellic acid effects on yield and quality of a seedless red table grape for saving irrigation water supply. Eur J Agron 2016; 80: 21-31.
[http://dx.doi.org/10.1016/j.eja.2016.06.015] , 13Crupi P, Bergamini C, Perniola R, Dipalmo T, Clodoveo ML, Antonacci D. A chemometric approach to identify the grape cultivar employed to pruduce nutraceutical fruit juice. Eur Food Res Technol 2015; 241: 487-96.
[http://dx.doi.org/10.1007/s00217-015-2478-y] , 24Liang Z, Owens CL, Zhong GY, Cheng L. Polyphenolic profiles detected in the ripe berries of Vitis vinifera germplasm. Food Chem 2011; 129(3): 940-50.
[http://dx.doi.org/10.1016/j. foodchem. 2011. 05.050] [PMID: 252123 22] ]. Apart from the first sampling date, the treatment with PhCO2 confirmed to be significantly related to ripening (p <0.001; F= 4.67). The concentrations of all the anthocyanins enhanced, especially as evident in the case of peonidin-3O-glucoside (3000 vs 1600 μg/g), cyanidin-3O-glucoside (290 vs 31 μg/g), and malvidin-3O-glucoside (140 vs 39 μg/g), which reached values from 2 to almost 10 folds higher in T2 than T1 grapes at the last sampling date (Table 1).
Since the analyses were conducted on extracts from a fixed weight of grounded dry skins, these outcomes allowed supposing that the different concentrations of the compounds were probably due to the alteration of their biosynthetic pathway and not the simple variation of berry dimensions and/or skin/flesh proportion [25Roby G, Harbertson JF, Adams DA, Matthews MA. Berry size and vinewater deficits as factors in winegrape composition: Anthocyanins and tannins. Aust J Grape Wine Res 2004; 10: 100-7.
[http://dx.doi.org/10.1111/j.1755- 0238. 2004. tb00012. x] , 26Suriano S, Alba V, Di Gennaro D, Suriano MS, Savino M, Tarricone L. Genotype/rootstocks effect on the expression of anthocyanins and flavans in grapes and wines of Greco Nero n. (Vitis vinifera L.). Sci Hortic (Amsterdam) 2016; 209: 309-15.
[http://dx.doi.org/10.1016/j. scienta. 2016. 07. 004] ]. For instance, the two pairs of anthocyanins, peonidin-3O-glucoside/cyanidin-3O-glucoside and malvidin-3O-glucoside/malvidin-3O-p-coumaroyl-glucoside, are biosynthetically linked by the actions of methyltransferases and p-coumaroyl-transferases, respectively [27He F, Mu L, Yan GL, et al. Biosynthesis of anthocyanins and their regulation in colored grapes. Molecules 2010; 15(12): 9057-91.
[http://dx.doi.org/10.3390/molecules15129057] [PMID: 21150825] ]. Therefore, it could be inferred that PhCO2 inhibited the methoxylation of a hydroxyl group of cyanidin to peonidin and, conversely, triggered the esterification of the glucoside moiety favoring the enzymatic conversion of malvidin-3O-glucoside to the more stable malvidin-3O-p-coumaroyl-glucoside, as significantly shown at the last sampling date (Fig. 3 ). These changes have also impacted both the hue and color stability of grapes, which are specifically influenced by the hydroxylation/methoxylation pattern of the B ring (Fig. 2
). These changes have also impacted both the hue and color stability of grapes, which are specifically influenced by the hydroxylation/methoxylation pattern of the B ring (Fig. 2 ) as well as the p-coumaroylation of glycosides in anthocyanins [27He F, Mu L, Yan GL, et al. Biosynthesis of anthocyanins and their regulation in colored grapes. Molecules 2010; 15(12): 9057-91.
) as well as the p-coumaroylation of glycosides in anthocyanins [27He F, Mu L, Yan GL, et al. Biosynthesis of anthocyanins and their regulation in colored grapes. Molecules 2010; 15(12): 9057-91.
[http://dx.doi.org/10.3390/molecules15129057] [PMID: 21150825] , 28Brar HS, Singh Z, Swinny E, Cameron I. Girdling and grapevine leafroll associated viruses affect berry weight, colour development and accumulation of anthocyanins in ‘Crimson Seedless’ grapes during maturation and ripening. Plant Sci 2008; 175: 885-97.
[http://dx.doi.org/10.1016/j.plantsci. 2008. 09. 005] ], indeed the skins of T2 ‘Crimson Seedless’ appeared darker red than T1 ones (Fig. 4 ).
).
Indeed, except for the case of h values, which were not significantly different, the PhCO2 treatment provoked a consistent decrease of L* as well as an increase of C*, which measure the lightness and purity of the color in T2 grapes, respectively. These t-test findings, graphically discussed through Box & Whisker plots, suggested a valuable shift toward more intense red berries as a consequence of the harpin proteins activity (Fig. 4 ).
).
CONCLUSION
In conclusion, the foliar treatment with harpin proteins was able to trigger the anthocyanins biosynthesis, during the ripening of ‘Crimson Seedless’. The concentrations of peonidin-3O-glucoside, cyanidin-3O-glucoside, and malvidin-3O-glucoside were particularly enhanced, up to 10 folds, in treated grapes at maturity. The color of berry skins was actually improved at harvest, too.
Therefore, the findings of this study are major arguments in favor of using PhCO2 as a valid alternative PGR for overcoming the issue of pigmentation and guarantying a higher level of bioactive compounds to this table grape variety.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
HUMAN AND ANIMAL RIGHTS
No animals/humans were used for studies that are the basis of this research.
CONSENT FOR PUBLICATION
Not applicable.
AVAILABILITY OF DATA AND MATERIALS
Not applicable.
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.
SUPPLEMENTARY MATERIAL
Supplementary material is available on the publishers Website along with the published article.
REFERENCES
| [1] | Peppi MC, Fidelibus MW, Dokoozliam N. Application timing and concentration of abscisic acid affect the quality of ‘Redglobe’ grapes. J Hortic Sci Biotechnol 2007; 82: 304-10. [http://dx.doi.org/10.1080/14620316.2007.115122 33] |
| [2] | Lurie S, Ovadia R, Nissim-Levi A, et al. Abscisic acid improves colour development in ‘Crimson Seedless’ grapes in the vineyard and on detached berries. J Hortic Sci Biotechnol 2009; 84: 639-44. [http://dx.doi.org/10.1080/14620316.2009.11512579] |
| [3] | Guidoni S, Hunter JJ. Anthocyanin profile in berry skins and fermenting must/wine, as affected by grape ripeness level of Vitis vinifera cv. Shiraz/R99. Eur Food Res Technol 2012; 235: 397-408. [http://dx.doi.org/10.1007/s00217-012-1744-5] |
| [4] | Carrieri C, Milella RA, Incampo F, et al. Antithrombotic activity of 12 table grape varieties. Relationship with polyphenolic profile. Food Chem 2013; 140(4): 647-53. [http://dx.doi.org/10.1016/j.foodchem.2012.10.132] [PMID: 236927 48] |
| [5] | Phan MAT, Bucknall MP, Arcot J. Interferences of anthocyanins with the uptake of lycopene in Caco-2 cells, and their interactive effects on anti-oxidation and anti-inflammation in vitro and ex vivo. Food Chem 2019; 276: 402-9. [http://dx.doi.org/10.1016/j.foodchem.2018.10.012] [PMID: 30409 612] |
| [6] | Popovic-Djordjevic J, Pejin B, Dramicanin A, et al. Wine chemical composition and radical scavenging activity of some cabernet franc clones. Curr Pharm Biotechnol 2017; 18(4): 343-50. [http://dx.doi.org/10.2174/1389201018666170313100919] [PMID: 28 294060] |
| [7] | Đorđević NO, Todorović N, Novaković IT, et al. Antioxidant activity of selected polyphenolics in yeast cells: The case study of Montenegrin Merlot Wine. Molecules 2018; 23(8): 1971. [http://dx.doi.org/10.3390/molecules23081971] [PMID: 300872 28] |
| [8] | Gagné S, Cluzet S, Mérillon JM, Gény L. ABA initiates anthocyanin production in grape cell cultures. J Plant Growth Regul 2011; 30: 1-10. [http://dx.doi.org/10.1007/s00344-010-9165-9] |
| [9] | Ferrara G, Mazzeo A, Matarrese AMS, et al. Ethephon as a potential abscission agent for table grapes: Effect on pre-harvest abscission, fruit quality, and residue. Front Plant Sci 2016; 7: 620. [http://dx.doi.org/10.3389/fpls.2016.00620] [PMID: 273034 07] |
| [10] | Crupi P, Antonacci D, Savino M, Genghi R, Perniola R, Coletta A. Girdling and gibberellic acid effects on yield and quality of a seedless red table grape for saving irrigation water supply. Eur J Agron 2016; 80: 21-31. [http://dx.doi.org/10.1016/j.eja.2016.06.015] |
| [11] | Vujović D, Maletić R, Popović-Đorđević J, Pejin B, Ristić R. Viticultural and chemical characteristics of Muscat Hamburg preselected clones grown for table grapes. J Sci Food Agric 2017; 97(2): 587-94. [http://dx.doi.org/10.1002/jsfa.7769] [PMID: 270982 41] |
| [12] | Spayd SE, Tarara JM, Mee DL, Ferguson JC. Separation of sunlight and temperature effects on the composition of Vitis vinifera cv. Merlot berries. Am J Enol Vitic 2008; 53: 171-82. |
| [13] | Crupi P, Bergamini C, Perniola R, Dipalmo T, Clodoveo ML, Antonacci D. A chemometric approach to identify the grape cultivar employed to pruduce nutraceutical fruit juice. Eur Food Res Technol 2015; 241: 487-96. [http://dx.doi.org/10.1007/s00217-015-2478-y] |
| [14] | Alfano JR, Collmer A. The type III (Hrp) secretion pathway of plant pathogenic bacteria: Trafficking harpins, Avr proteins, and death. J Bacteriol 1997; 179(18): 5655-62. [http://dx.doi.org/10.1128/jb.179.18.5655-5662.1997] [PMID: 92944 18] |
| [15] | Li M, Yu ML, Zhang ZQ, Liu QN, Wu YC, Liu ZG. Effects of Harpin protein on fruit quality and shelf life of winter jujube (Ziziphus jujube Mill. Cv. Dongzao). Biol Agric Hortic 2013; 29: 58-68. [http://dx.doi.org/10.1080/01448765.2012.762729] |
| [16] | Liu R, Lü B, Wang X, et al. Thirty-seven transcription factor genes differentially respond to a harpin protein and affect resistance to the green peach aphid in Arabidopsis. J Biosci 2010; 35(3): 435-50. [http://dx.doi.org/10.1007/s12038-010-0049-8] [PMID: 20826953] |
| [17] | Akbuk N, Tezcan H, Akbudak B, Seniz V. The effect of harpin protein on plant growth parameters, leaf chlorophyll, leaf colour and percentage rotten fruit of pepper plants inoculated with Botrytis cinerea. Sci Hortic (Amsterdam) 2006; 109: 107-12. [http://dx.doi.org/10.1016/j.scienta.2006.03.008] |
| [18] | Lorenz DH, Eichhorn KW, Blei-holder H, Klose R, Meier U, Weber E. Phänologische Entwicklungsstadien der Weinrebe (Vitis vinifera L.ssp. vinifera). Vitic Enol Sci 1994; 49: 66-70. |
| [19] | Office International de la Vigne et du Vin. 1990. |
| [20] | Ferrara G, Mazzeo A, Matarrese AMS, et al. Application of abscisic acid (S-ABA) to ‘Crimson Seedless’ grape berries in a Mediterranean climate: Effects on color, chemical characteristics, metabolic profile, and S-ABA concentration. J Plant Growth Regul 2013; 32: 491-505. [http://dx.doi.org/10.1007/s003 44- 012- 9316-2] |
| [21] | Vujović D, Pejin B, Popović Djordjević J, Velicković M, Tesević V. Phenolic natural products of the wines obtained from three new Merlot clone candidates. Nat Prod Res 2016; 30(8): 987-90. [http://dx.doi.org/10.1080/14786419.2015.1079191] [PMID: 263085 89] |
| [22] | Ferrara G, Mazzeo A, Matarrese AMS, et al. Application of abscisic acid (S-ABA) and sucrose to improve colour, anthocyanin content and antioxidant activity of cv. Crimson Seedless grape berries. Aust J Grape Wine Res 2015; 21: 18-29. [http://dx.doi.org/10.1111/ ajgw.1211 2] |
| [23] | Singh Brar H, Singh Z, Swinny E. Dynamics of anthocyanin and flavonol profiles in the ‘Crimson Seedless’ grape berry skin during development and ripening. Sci Hortic (Amsterdam) 2008; 117: 349-56. [http://dx.doi.org/10.1016/j.scienta.2008.05.007] |
| [24] | Liang Z, Owens CL, Zhong GY, Cheng L. Polyphenolic profiles detected in the ripe berries of Vitis vinifera germplasm. Food Chem 2011; 129(3): 940-50. [http://dx.doi.org/10.1016/j. foodchem. 2011. 05.050] [PMID: 252123 22] |
| [25] | Roby G, Harbertson JF, Adams DA, Matthews MA. Berry size and vinewater deficits as factors in winegrape composition: Anthocyanins and tannins. Aust J Grape Wine Res 2004; 10: 100-7. [http://dx.doi.org/10.1111/j.1755- 0238. 2004. tb00012. x] |
| [26] | Suriano S, Alba V, Di Gennaro D, Suriano MS, Savino M, Tarricone L. Genotype/rootstocks effect on the expression of anthocyanins and flavans in grapes and wines of Greco Nero n. (Vitis vinifera L.). Sci Hortic (Amsterdam) 2016; 209: 309-15. [http://dx.doi.org/10.1016/j. scienta. 2016. 07. 004] |
| [27] | He F, Mu L, Yan GL, et al. Biosynthesis of anthocyanins and their regulation in colored grapes. Molecules 2010; 15(12): 9057-91. [http://dx.doi.org/10.3390/molecules15129057] [PMID: 21150825] |
| [28] | Brar HS, Singh Z, Swinny E, Cameron I. Girdling and grapevine leafroll associated viruses affect berry weight, colour development and accumulation of anthocyanins in ‘Crimson Seedless’ grapes during maturation and ripening. Plant Sci 2008; 175: 885-97. [http://dx.doi.org/10.1016/j.plantsci. 2008. 09. 005] |







