- Home
- About Journals
-
Information for Authors/ReviewersEditorial Policies
Publication Fee
Publication Cycle - Process Flowchart
Online Manuscript Submission and Tracking System
Publishing Ethics and Rectitude
Authorship
Author Benefits
Reviewer Guidelines
Guest Editor Guidelines
Peer Review Workflow
Quick Track Option
Copyediting Services
Bentham Open Membership
Bentham Open Advisory Board
Archiving Policies
Fabricating and Stating False Information
Post Publication Discussions and Corrections
Editorial Management
Advertise With Us
Funding Agencies
Rate List
Kudos
General FAQs
Special Fee Waivers and Discounts
- Contact
- Help
- About Us
- Search

The Open Clinical Cancer Journal
(Discontinued)
ISSN: 1874-1894 ― Volume 5, 2011
Oncolytic Effects of the E Variant of Encephalomyocarditis Virus to Human Neuroblastoma in in Vitro and in Vivo
Masazumi Adachi*, 1, 2, Steven E Brooks1, Maxine R. Stein1, Bernice E Franklin1, Francis A Caccavo3
Abstract
The oncolytic effects of encephalomyocarditis (EMC) virus on of human neuroblastoma (NB) were studied in vitro employing NB cell cultures and in vivo employing tumors grown in nude mice and nude rats. In short-term studies, human NB cell (HTB10) cultures were exposed to the virus 104 Tissue Culture Infectious Doses (TCIDs). The TCIDs were used to titer the maximum effect of EMC virus on L-929 cells. In vitro studies showed 100% Cytopathic Effect (CPE) at 48 hours. The CPE was used to observe pathologic effects of the cells. The in vivo studies showed necrosis and degeneration in the tumors grown in nude mice at 7 days following treatment with the virus. The nude mice exhibited a high mortality rate after exposure to the virus. The long-term studies were performed on tumors grown in nude rats, since they were more tolerant to the viral treatment. On the 58th day after the viral treatment of tumor bearing rats, 8 out of the 12 tumors had disappeared, and the remaining 4 tumors were grossly necrotic. The average size of the infected tumors was 2.13 cm2 as compared with 30.5 cm2 in the control. Microscopically the treated tumors showed severe necrosis and microcystic degeneration. The observed gross decrease in treated tumor size together with the presented light and electron microscopic studies, support the potential value of viral treatment of neuroblastoma.
Article Information
Identifiers and Pagination:
Year: 2009Volume: 3
First Page: 1
Last Page: 9
Publisher Id: TOCCJ-3-1
DOI: 10.2174/1874189400903010001
Article History:
Received Date: 20/10/2008Revision Received Date: 30/7/2009
Acceptance Date: 22/8/2009
Electronic publication date: 26/11/2009
Collection year: 2009
open-access license: This is an open access article licensed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted, non-commercial use, distribution and reproduction in any medium, provided the work is properly cited.
* Address correspondence to this author at the Department of Laboratories, Kingsbrook Jewish Medical Center USA, 585 Schenectady Ave., Brooklyn, N.Y. 11203, USA; Tel: (347) 804-6920; Fax: (718) 604-5777; E-mail:madachi@kingsbrook.org, fcaccavo@health.nyc.gov
| Open Peer Review Details | |||
|---|---|---|---|
| Manuscript submitted on 20-10-2008 |
Original Manuscript | Oncolytic Effects of the E Variant of Encephalomyocarditis Virus to Human Neuroblastoma in in Vitro and in Vivo | |
INTRODUCTION
Encephalomyocarditis (EMC) virus is in the family of picornavirus. The virion is 20 to 30 nm in diameter and it consists of a capsid shell of 60 subunits arranged with icosahedral symmetry around a genome that has 11 variants [1]. The virus has been investigated at our laboratory for many years [1-2]. The E variant (EMC-E) is highly neurotropic [1-5,7,9-1,13] and usually not pathogenic to man [1,14]. The present investigation shows the oncolytic effects of the EMC-E virus on human neuroblastoma.
Neuroblastoma (NB) is one of the most frequently encountered malignant tumors in children. About 83% of cases are detected in children under the age of six [15]. At the time of diagnosis, the majority of patients (almost 80%) have been reported to show metastasis [16]. Therefore, this virus mediated therapy of the target cells suggests a possibly effective way to treat this malignant tumor.
MATERIALS AND METHODS
Human NB cells (HTB10) were obtained from the American Type Culture Collection. The cells were main tained in T-30 flasks in Minimum Essential Medium (MEM) containing 10% fetal bovine serum (FBS). The EMC-E virus was prepared by infecting the monolayer of L-929 cells with the stock virus for 48 hours until cytopathic effect (CPE) [7] was evident. The CPE was used to observe maximum pathologic effect. The cells and supernatant media were collected and subjected to 3 cycles of freeze-thaw. The suspension was centrifuged at 2000 x g to remove cell debris, and the supernatant fluid containing the virus was frozen in liquid nitrogen in 1 ml aliquots. The virus suspension was titered on the monolayer of L-929 cells using the CPE endpoint technique. For in vitro studies, the NB cells were grown to 80% confluence in T-30 falcon flasks. The cultures were washed with phosphate buffered saline (Dulbeccos) and infected with 104 Tissue Culture Infectious Doses (TCIDs) of the EMC-E virus in 1 ml of MEM containing 10% FBS. The control cultures were handled in a similar manner but were not exposed to the virus. The control and experimental cultures were harvested at 16, 20, 24, 32, 40, 48 and 72 hours post-infection and processed for light and electron microscopic studies. In the in vivo studies, tumors of the NB cells were established in nude mice and nude rats by injecting 107 of the cells into the area of the right shoulder. After 3 to 6 weeks, animals exhibited progressively growing tumors at the site of inoculation, which were confirmed to be of human origin by karyotype analysis. From these animals, 12 nude mice and 25 nude rats were selected which had the tumors of approximately 1 cm in diameter. The experimental subgroup of 8 mice and 16 rats was injected in the left flank with 104 TCIDs of the EMC-E virus. The control group which was not inoculated with the virus consisted of the remaining 4 mice and 9 rats. In the short-term studies, the 8 experimental and 4 control mice were sacrificed on the 7th day post-viral infection. In the long-term studies, the rats were divided into two groups. The first group consisting of 4 experimental and 4 control rats was sacrificed on the 48th day post-viral infection. The second group consisting of 12 experimental and 5 control rats was scarified on the 58th day post-viral infection. The experiment was terminated on the 58th day because of the large size of tumors in the control group. The tumor tissues were prepared for light and electron microscopic studies. Ultrastructural procedures were performed according to our previous publication [17]. The animals were housed and handled in accordance with the guidelines of the National Research Council.
RESULTS
In the cell cultures, after the exposure to EMC-E virus, alterations of NB cells are seen at 20 hours (Fig. 1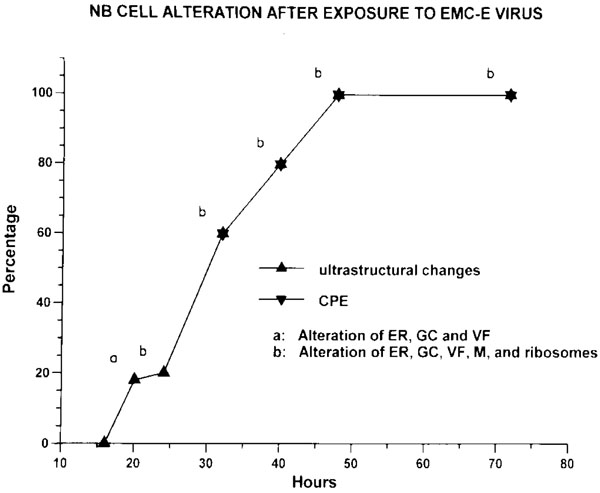 ).
).
The initial changes were noted in the endoplasmic reticulum (ER), Golgi complex (GC) and vacuolar formation (VF). At 24 hours, ribosomes were markedly decreased in addition to the changes of ER, GC and VF. The cultures showed 60% CPE at 32 hours and 100% CPE at 48 hours. The control cultures showed numerous small cells with the round to ovoid nuclei. One of the cultures at 32 hours is seen in Fig. (2A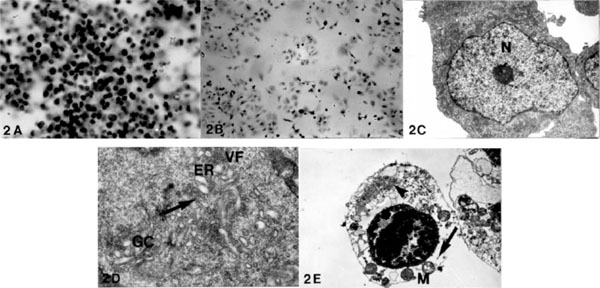 ) as compared with a marked reduction of the treated cells at the same period in Fig. (2B
) as compared with a marked reduction of the treated cells at the same period in Fig. (2B ). The ultrastructure of the control cells consisted of the large nuclei with the prominent nucleoli and a small amount of the cytoplasm with preserved subcellular organelles (Fig. 2C
). The ultrastructure of the control cells consisted of the large nuclei with the prominent nucleoli and a small amount of the cytoplasm with preserved subcellular organelles (Fig. 2C ). The infected cells at 20 hours showed dilated ER and GC, and VF were noted from ER (Fig. 2D
). The infected cells at 20 hours showed dilated ER and GC, and VF were noted from ER (Fig. 2D ). At 24 hours, a marked reduction of the ribosomes was observed (Fig. 2E
). At 24 hours, a marked reduction of the ribosomes was observed (Fig. 2E ) in addition to the changes of ER, GC and VF.
) in addition to the changes of ER, GC and VF.
In the short-term in vivo studies, the gross changes of the infected and control tumors at 7 days are noted in Fig. (3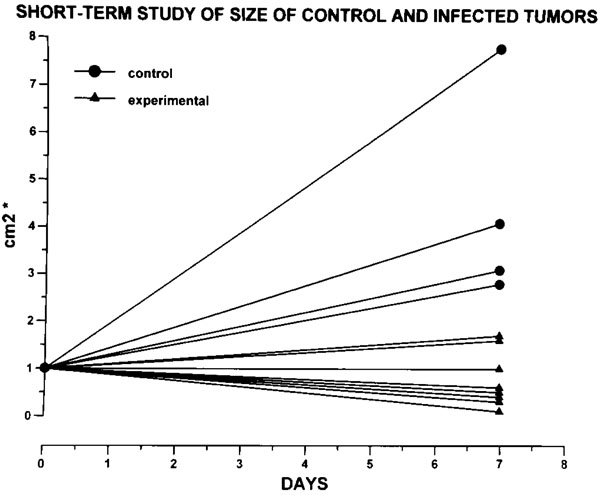 ).
).
 |
Fig. (3) The size of the tumors in the short-term study is expressed by multiplications of the length and width that correspond to the longterm
studies (see Fig. 7A |
In the treated mice tumors ranged from 0.1 cm2 to 1.7 cm2 (average: 0.77 cm2; sd: 0.59 cm2) as compared with the control tumors which ranged from 2.8 cm2 to 7.8 cm2 (average: 4.9 cm2, sd: 2.30 cm2). The treated tumors were smaller and soft, while the controls were larger and firm. Example is shown in Fig. (4 ).
).
 |
Fig. (4) The treated tumor at 7 days on the right side and the control on the left side show that the experimental tumor is smaller and soft as compared with the larger and firm control. |
Histological studies showed that the control tumors were markedly cellular (Fig. 5A ) and revealed the preserved tumor cells in hemorrhagic areas (Fig. 5B
) and revealed the preserved tumor cells in hemorrhagic areas (Fig. 5B ). In contrast, the infected tumors were markedly necrotic (Fig. 5C
). In contrast, the infected tumors were markedly necrotic (Fig. 5C ) and showed severe degeneration with the pyknotic nuclei (Fig. 5D
) and showed severe degeneration with the pyknotic nuclei (Fig. 5D ). Ultra-structure of the control tumors at 7 days showed the large nuclei and a small amount of the cytoplasm with preserved subcellular organelles. (Fig. 5E
). Ultra-structure of the control tumors at 7 days showed the large nuclei and a small amount of the cytoplasm with preserved subcellular organelles. (Fig. 5E ), while the infected cells at the same interval showed severe degeneration with a marked loss of the ribosomes and single membrane bound vacuoles from the Golgi and ER (Fig. 5F
), while the infected cells at the same interval showed severe degeneration with a marked loss of the ribosomes and single membrane bound vacuoles from the Golgi and ER (Fig. 5F ).
).
To observe the longer-term effects of the oncolytic process, the first group of the tumor-bearing nude rats was studied at 48 days. The gross changes of the infected tumors are noted in Fig. (6A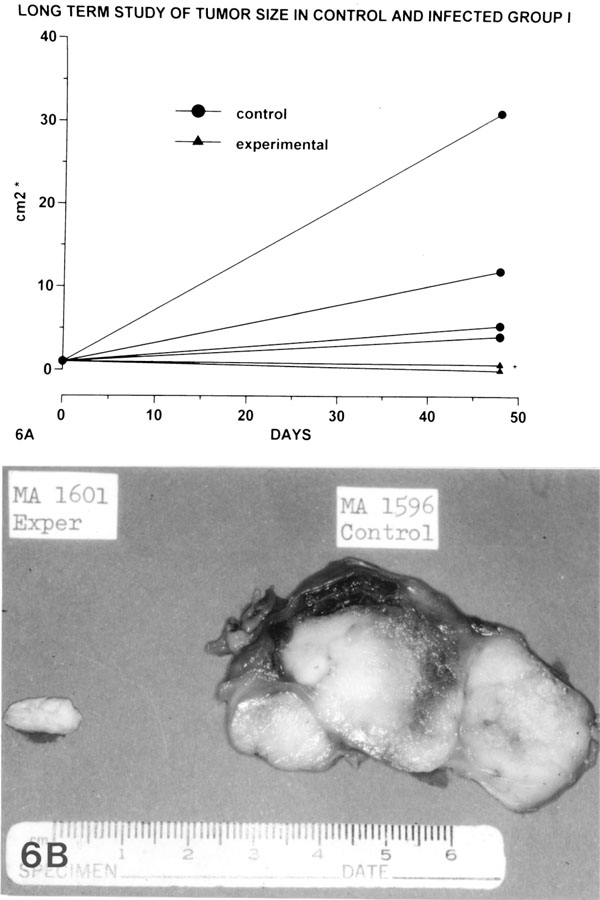 ). The size of infected tumors ranged from 0 to 0.7 cm2 (average: 0.52 cm2; sd: 0.35 cm2) as compared with the tumors in the controls which ranged from 4.1 cm2 to 31.2 cm2 (average: 13.17 cm2; sd: 12.50 cm2).
). The size of infected tumors ranged from 0 to 0.7 cm2 (average: 0.52 cm2; sd: 0.35 cm2) as compared with the tumors in the controls which ranged from 4.1 cm2 to 31.2 cm2 (average: 13.17 cm2; sd: 12.50 cm2).
One of the gross examples is observed in Fig. (6B ). The experimental tumor is small and totally necrotic, while the control tumor is massive and firm.
). The experimental tumor is small and totally necrotic, while the control tumor is massive and firm.
In order to increase the number of observation and to study the pattern of changes of the tumors for a longer duration, the second long-term studies group was carried out (Fig. 7 ). At 58 days, the size of control group tumors ranged from 12.9 cm2 to 57.1 cm2 (average: 30.5 cm2; sd: 17.99 cm2), while the infected group tumors ranged from 0 to 15.8 cm2 (average: 2.13 cm2; sd: 5.10 cm2). At the same period, eight out of the 12 treated tumors had totally disappeared, and the other two infected tumors were only trace (0.05 cm2). The remaining two tumors (15.8 cm2 and 9.6 cm2, respectively) were grossly necrotic. All experimental animals were healthy during the long-term study.
). At 58 days, the size of control group tumors ranged from 12.9 cm2 to 57.1 cm2 (average: 30.5 cm2; sd: 17.99 cm2), while the infected group tumors ranged from 0 to 15.8 cm2 (average: 2.13 cm2; sd: 5.10 cm2). At the same period, eight out of the 12 treated tumors had totally disappeared, and the other two infected tumors were only trace (0.05 cm2). The remaining two tumors (15.8 cm2 and 9.6 cm2, respectively) were grossly necrotic. All experimental animals were healthy during the long-term study.
Histological studies showed compactly arranged cells with frequent mitotic figures in the controls at 58 days (Fig. 8A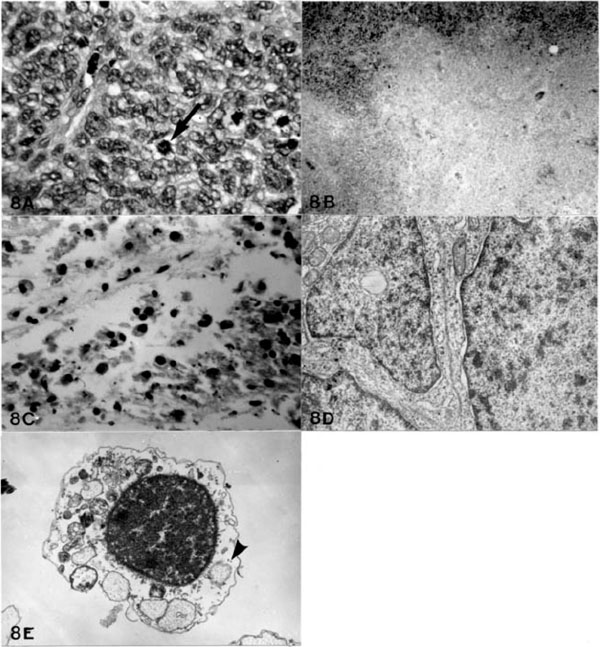 ), while the large infected tumors at the same period displayed severe necrosis (Fig. 8B
), while the large infected tumors at the same period displayed severe necrosis (Fig. 8B ) and microcystic changes with proliferation of macrophages (Fig. 8C
) and microcystic changes with proliferation of macrophages (Fig. 8C ). The infected tumors of trace size at the same period were also totally necrotic. Ultrastructure of the control tumors at 58 days revealed compactly arranged cells with large nuclei with small amount of the cytoplasm with preserved subcellular organelles (Fig. 8D
). The infected tumors of trace size at the same period were also totally necrotic. Ultrastructure of the control tumors at 58 days revealed compactly arranged cells with large nuclei with small amount of the cytoplasm with preserved subcellular organelles (Fig. 8D ). The infected cells at the same period showed a marked reduction of the ribosomes with severe degeneration of the subcellular organelles (Fig. 8E
). The infected cells at the same period showed a marked reduction of the ribosomes with severe degeneration of the subcellular organelles (Fig. 8E ).
).
DISCUSSION
In addition to their clinical data, molecular biology and histopathological typing of the patients predict the prognosis of NB [18-23]. Studies have shown that infants under the age of 18 months have a good clinical course, if they have a low clinical stage, hyperdiploid DNA-content and the expression of the c-trk proto-oncogene related with the high affinity NGF (nerve growth factor) receptor. On the other hand, children more than 2 year of age, with higher clinical stage, near diploid DNA-content, chromosome 1p deletions, N-myc gene amplification and the absence or low expression of the c-trk gene have a poor clinical course [23-28]. Signs and symptoms of the disease are related to the original site and spread of the tumor. The majority (63-76%) of tumors originated from the adrenal medulla and sympathetic ganglia [15,16, 24-26]. Mediastinal and abdominal masses create respiratory distress and complications of the urinary system. Neurological symptoms are also observed by compression and autoantibodies produced against the tumor [28-30]. Systemic symptoms, such as hypertension, coagulopathies and diarrhea are associated with functional hormone secretion [31-36]. Since 90% to 95% of the tumor produce catecholamines in urine, analysis of the urinary metabolites is important in addition to performing diagnostic procedures, such as computerized tomography, magnetic resonance imaging, ultrasonography and fine-needle aspiration biopsy for the diagnosis at the early stage [21, 23, 24, 37-39].
The therapeutic choices for the disease are surgery, radiation, chemotherapy and bone marrow transplantation according to clinical genetic subtypes of the tumor [21]. However, long-term control is seldom achieved since the tumor has a tendency to produce widespread metastases. In these cases, the oncolytic effects of the EMC-E virus is an ideal approach to treat desired target cells based on the virus special affinity for the tumor receptors. Specific molecular arrangements on the viral surface are complementary to the receptors on the target cells. These interactions are well demonstrated by the investigations of enteroviruses and coronaviruses [40-43]. Further details of the virus-receptor interactions have been described [44-49].
The EMC virus causes a high mortality rate in mice [1,2,10,11,13]. However, an extensive clinical study has shown that this rodent virus is not implicated in any significant pathogenesis in man [1,14]. Isolation and serologic studies of large numbers of patients with meningitis and encephalitis failed to implicate this rodent virus in human disease. The structure and functional details of picornaviruses have been demonstrated [44, 50]. During a process of the entry of the virus into cell membranes, sequential changes of the virion polypeptides occur, and interaction of the virion with specific receptors dissociates the viral polypeptides in order to permit release of the viral RNA in the host cytoplasm [45, 51]. After the entry of the virus in the host cells, picornaviruses inhibit synthesis of host protein and direct the cells to synthesize viral proteins [52, 53].
Our current long-term studies have shown the 100% cure rate (total disappearance: 10 out of 12 cases; trace size and total necrosis: 2 cases) as compared with the control (average size: 30.5 cm2). These results suggest the concept that utilizing the EMC-E virus as an oncolytic agent may be a new effective way of virotechnology (virotech) to approach the treatment of this tumor and other malignant tumors of the nervous system.






